��Ŀ����
���й�����Һ��˵����ȷ���ǣ� ��
A�����ܽ��������ʵ����ʽ����ܼ� B�����Ǿ�һ�ġ��ȶ���Һ��һ������Һ
C����Һһ������ɫ���� D��������Һһ����Ũ��Һ
A ��������A�����ܽ��������ʵ����ʽ����ܼ� ����A��ȷ��B�����Ǿ�һ�ġ��ȶ��Ļ����һ������Һ����B����C����Һһ���Ǿ�һ�ġ��ȶ��ģ�����һ����ɫ���ġ���C����D��������Һ��һ����Ũ��Һ����D������Ӧ�����ǵ��ճ���������������еĹ�ϵ��
�������˵�θ����_______�����θҺ���ڹ��������θ�������ú�������������ҩ��ɻ��ⲡʹ���䷴Ӧ�Ļ�ѧ����ʽΪ__________________________________________��
�� ����ͷ��������ʱ�������������л��ᣬ����ͷǰ��ӽ������Ĵ����С�մ��
�������ʣ���������ò�����__________����(�ѧʽ)��ʹ��������ͷ���ɶ�ף�������ļ������ʹ��࣬�������Ż������еμ�������____________(�ѧʽ)�����Ե�ζ�ϣ�������������ͷ���ɬ����ɫ���ơ�
�� δ�����ˮ��������ɬζ��������Ϊˮ���ﺬ�����ᡣ��ɬ�ķ���֮һ�ǣ�����������ˮ����ʯ�һ���ʯ���飬�ñ仯�Ļ�ѧ����ʽ�� ___________________________ ��Ȼ���������ˮϡ�ͣ�ȡ�ϲ���ҹ������ˮ��5��6�켴�ɳ�ȥɬζ��
���˱����涣ҧ��Ƥ���������� ����������������Ƥ����ע���������������¡��������ұ����е������Ʒ_________��__________(����)Ϳ�ڱ�ҧ��Ƥ���ϣ�ʹ�������ʧ��
���� | A | B | C | D |
����ˮ | ʳ�� | ���� | ʳ��ˮ | |
pH | 10 | 3 | 9 | 7 |
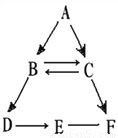
�� �����پٳ�һ�������������г�����������������____________________________��
���� Al��OH��3+3HCl�TAlCl3+3H2O CO2 CH3COOH CaO+H2O�TCa��OH��2 A C �������Ƹ����������� ����������1���˵�θҺ�к������ᣬ�����ܺ�����������Ӧ�����Ȼ�����ˮ��θҺ���ڹ��������θ�������ú�������������ҩ��ɻ��ⲡʹ����ѧ����ʽΪAl��OH��3+3HCl�TAlCl3+3H2O�� ��2��������������ʷ�Ӧ�����ɶ�����̼���壬ʹ��������ͷ��...�±��У���ȥ�����������ʵķ�������ȷ���ǣ� ��
ѡ�� | ���� | �������� | ��ȥ���ʵķ��� |
A | H2 | HCl���� | ͨ��NaCH��Һ����ͨ��Ũ���� |
B | NaCl��Һ | Na2CO3 | ��������ϡ���� |
C | CO | CO2 | ͨ�����ȵ�Fe2O3 |
D | CaCO3��ĩ | KCl | ��ˮ�ܽ⡢���ˡ����� |