��Ŀ����
9�� ij��ѧ��ȤС��ʹ����ͼ��ʾװ�ã���ij����ͭ�Ͻ�ijɷֽ��в�������ȡ����ϡ�������ձ��У��������м���14.0g�Ͻ���Ʒ��ʼ��ʱ������������ƽ�Ķ�����¼�ڱ��У���ش��������⣺
ij��ѧ��ȤС��ʹ����ͼ��ʾװ�ã���ij����ͭ�Ͻ�ijɷֽ��в�������ȡ����ϡ�������ձ��У��������м���14.0g�Ͻ���Ʒ��ʼ��ʱ������������ƽ�Ķ�����¼�ڱ��У���ش��������⣺| ��¼ʱ�� | ���ձ� | ���� ����� | �������ۺ� 5���� | �������ۺ� 12���� | �������ۺ� 21���� | �������ۺ� 40���� | �������ۺ� 3Сʱ |
| ������g�� | 28.5 | 169.7 | 183.6 | 183.5 | 183.4 | 183.3 | 183.3 |
��2����Ӧ��������������Ϊ0.4g��
��3������Ͻ�����Ԫ�ص������ж��ٿˣ���ȫ��Ӧ���ձ����ε���������Ϊ���٣�
���� ���ݱ����ṩ��������������������������ٸ��ݻ�ѧ����ʽ�������������ط��������
��� �⣺��1����Ϊ�����Ũ��Խ��ӦԽ�죬���ŷ�Ӧ�Ľ��У������Ũ�ȼ�С����Ӧ�ٶȱ��������������������ٶ��ɿ������
��2������������������169.7g+14.0g-183.3g=0.4g
��3����Ͻ���Fe������Ϊx�����ɵ�FeSO4����Ϊy��
Fe+H2SO4=FeSO4+H2��
56 152 2
x y 0.4g
$\frac{56}{x}=\frac{152}{y}=\frac{2}{0.4g}$
x=11.2g��y=30.4g��
��Һ������Ϊ��11.2g+��169.7g-28.5g��-0.4g=152g��
������������������Ϊ$\frac{30.4g}{152g}$��100%=20%��
�𰸣�
��1����Ϊ�����Ũ��Խ��ӦԽ�죬���ŷ�Ӧ�Ľ��У������Ũ�ȼ�С����Ӧ�ٶȱ��������������������ٶ��ɿ������
��2��0.4��
��3���Ͻ�����Ԫ�ص�����Ϊ11.2g��������������������Ϊ20%��
���� ���ݱ����ṩ����Ϣ�����жϷ�Ӧ���е��������������ƽ�������ٱ仯ʱ��˵���Ѿ���ȫ��Ӧ������������������������������������һ�����������������

 ��У����ϵ�д�
��У����ϵ�д�
| A�� | ��CuCl2��NH4Cl������ | B�� | ��Fe����������� | ||
| C�� | ��Fe��NaCl��C��KNO3������ | D�� | ��CO2��Cu������ |
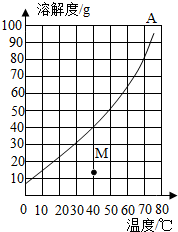 ��ͼ��A���ʵ��ܽ������ͼ�������B�����ڲ�ͬ�¶�ʱ���ܽ�ȣ�
��ͼ��A���ʵ��ܽ������ͼ�������B�����ڲ�ͬ�¶�ʱ���ܽ�ȣ� | �¶�/�� | 0 | 20 | 40 | 60 | 80 |
| B�����ܽ��/g | 32 | 35 | 40 | 45 | 55 |
��2��40��ʱ��A��B�������ʵı�����Һ�����ʵ�����������ȣ�
��3����Ҫ��A����Һ����ȡA���ʣ�С��ͬѧ��ΪҪ�������ܼ�ˮ�ķ�����С��ͬѧ��ΪҪ����ȴ�ȱ�����Һ�ᾧ�ķ���������ΪС��ͬѧ�ķ���������
��4��40��ʱ����50gA���ʼ���100gˮ�У�����ܽ���γ���Һ������Ϊ140g��

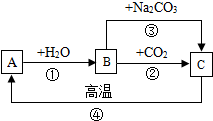 A��B��C�����ֳ����ĺ��ƻ��������֮������ͼ��ʾ��ת����ϵ�����ֲ�����ȥ����
A��B��C�����ֳ����ĺ��ƻ��������֮������ͼ��ʾ��ת����ϵ�����ֲ�����ȥ����

 ��֪A��B��C��D��E��Ϊ���л�ѧ�������ʣ����У�Eͨ��Ϊ���壬�����ת���Ĺ�ϵ��ͼ��ʾ��������������ش��й����⣺
��֪A��B��C��D��E��Ϊ���л�ѧ�������ʣ����У�Eͨ��Ϊ���壬�����ת���Ĺ�ϵ��ͼ��ʾ��������������ش��й����⣺