��Ŀ����
17����ҵ���õ��NaCl��Һ�ķ�������ȡNaOH��Cl2��H2����Ӧ�Ļ�ѧ����ʽΪ��2NaCl+2H2O$\frac{\underline{\;ͨ��\;}}{\;}$2NaOH+H2��+Cl2������ȡһ������������NaCl��Һ87.3g���е�⣬��NaCl��ȫ��Ӧʱ������0.2gH2����1����NaCl��ȫ��Ӧʱ������Cl2������Ϊ7.1g���μӷ�Ӧ��NaCl������Ϊ11.7g��
��2����NaCl��ȫ��Ӧʱ������NaOH��Һ�����ʵ���������Ϊ���٣����������ɵ�Cl2��H2ȫ���ݳ�����Ҫ�м�����̣�
���� ���ݷ�Ӧ�ķ���ʽ��������������������ɵ��������������������Ͳμӷ�Ӧ��NaCl���������ٸ������������ļ��㹫ʽ���㼴�ɣ�
��� �⣺��1��������Cl2������Ϊx���μӷ�Ӧ��NaCl������Ϊy�������������Ƶ�����Ϊz
2NaCl+2H2O$\frac{\underline{\;ͨ��\;}}{\;}$2NaOH+H2��+Cl2��
117 80 2 71
z y 0.2g x
$\frac{117}{z}=\frac{80}{y}=\frac{2}{0.2g}=\frac{71}{x}$
��ã�x=7.1g y=8g z11.7g
��2������NaOH��Һ�����ʵ���������Ϊ��$\frac{8g}{87.3g-0.2g-7.1g}��100%$=10%
�ʴ�Ϊ����1��7.1��11.7����2������NaOH��Һ�����ʵ���������Ϊ10%��
���� ���������û�ѧ����ʽ�ļ����⣬��Ϊ��������ķ���ʽ�Ѿ�������ʹ�ѶȽ����˺ܶ࣬����Ҫ����������������Ĺ�ʽ��

��ϰ��ϵ�д�
 Сѧ�̲�ȫ��ϵ�д�
Сѧ�̲�ȫ��ϵ�д� Сѧ��ѧ������ѿڶ���ϵ�д�
Сѧ��ѧ������ѿڶ���ϵ�д� ������Ӧ�������������ϵ�д�
������Ӧ�������������ϵ�д� �㽭֮�ǿ�ʱ�Ż���ҵϵ�д�
�㽭֮�ǿ�ʱ�Ż���ҵϵ�д�
�����Ŀ
8���������ǻ�ѧѧϰ�г��õ�˼ά����������������ȷ���ǣ�������
| A�� | �кͷ�Ӧ�����κ�ˮ�����������κ�ˮ�ķ�Ӧһ�����кͷ�Ӧ | |
| B�� | ���������ɲ�ͬ��Ԫ����ɵĴ���������ɲ�ͬ��Ԫ����ɵĴ�����һ���ǻ����� | |
| C�� | ������ֻ����һ��Ԫ�أ�����ֻ����һ��Ԫ�ص�����һ���ǵ��� | |
| D�� | ��ͬһ�������У�����Ԫ�������ۣ����Էǽ���Ԫ��һ���Ը��� |
5�������к��������ڹ�����������������������������������ɱ������̣�������Ϊ��������
| A�� | ����֮���м�϶ | B�� | �����ڲ����˶� | C�� | ���Ӻ�С | D�� | �����ǿɷֵ� |
2���������ʵ���;������ǣ�������
| A�� |  ������������ | B�� |  ������ϩ����ʳƷ��װ | ||
| C�� |  �ɱ������˹����� | D�� |  ϡ������������� |
9��ʵ������̼��غ�̼��ƵĹ�������75g��ʹ֮��500g��������Ϊ14.6%�������ַ�Ӧ������Ӧ�����Һ���ɵõ�82.7g���壮��ԭ������н���Ԫ�ص���������Ϊ��������
| A�� | 44% | B�� | 46% | C�� | 50% | D�� | 55% |
6�����л�ѧʵ�������ȷ���ǣ�������
| A�� |  ȡ��ʳ�� | B�� |  �������� | C�� |  ����Һ�� | D�� |  ϡ��Ũ���� ϡ��Ũ���� |
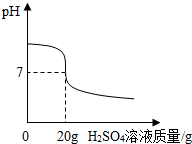 ijС������4.9%��ϡH2SO4��һƿ��ǩ�����NaOH��Һ������������
ijС������4.9%��ϡH2SO4��һƿ��ǩ�����NaOH��Һ������������