��Ŀ����
19���������Ϸ�չ��ʳƷ��ȫ���ⱻ����Խ��Խ���ӣ���1989�꣬����ΪʳƷ��ȾԴ֮һ������������֯����������ÿ�յ���������Ӧ������4�������£�ը����ʱ������뻯ѧʽΪKAl��SO4��2•12H2O[��Է���������474]�����������ɼ�����1000�˵��õ�ը���������У�������������2�ˣ���ش��������⣺��1���˴��������������ָ����A ������ĸ����
A����Ԫ�� B�������� C�����Ͻ� D��ֻ������
��2��KAl��SO4��2•12H2O����Ԫ�ص�������������������ȷ��O.1%����
��3����ÿ����������30�ˣ���ÿ�������к���Ԫ�ؼ����ˣ�������ը������������ģ�
���� ��1���������е�֪ʶ���з�������������Ԫ����ɵģ������к��е���Ԫ�أ�
��2��Ԫ�ص���������=$\frac{���ԭ��������ԭ�Ӹ���}{��Է�������}��100%$���ݴ˽��
��3������������Ԫ�������ļ��㷽����������
��� �⣺��1�������к��е���Ԫ�أ��˴�ʳ�����������ָ������Ԫ�أ����A��
��2����Ԫ�ص���������Ϊ��$\frac{27}{39+27+32��2+16��20+1��24}��100%$��5.7%�����5.7%��
��3��ÿ�������к���Ԫ�ص�����Ϊ��$\frac{2g}{1000g}��30g��5.7%$�T0.00342g=3.42mg�����3.42��
���� ���⿼���˻�ѧ�����彡����֪ʶ�Լ�����������жϺ��йصļ��㣬��ɴ��⣬�����������е�֪ʶ���У�

��ϰ��ϵ�д�
�����Ŀ
10����һ�죬ijѧϰС��Ұ�������������Ʒ���±���
��1���������в�ʳ��ʱ�����Ƿ�������ʳ�ﲻ����ƽ����ʳ��Ҫ����Ϊʳ����ȱ����ˮ�����߲ˣ�
��2��Ұ����У���һͬѧ���۷��������ˣ��۷䶾Һ�����ԣ�����ͬѧ�������÷���ˮΪ��ͿĨ���������֢״�����ᣬ������Ϊ����ˮ�ʼ��ԣ�
��3��Ұ��������С�ƴ������л�Ϥ��ijƷ�ƽ���ÿ100mL����Ӫ�����ı���������0.2g����Ӫ��������Ԫ����������Ϊ12.5%���ɴ˿ɼ���һƿ448mL��Ʒ�������������൱�ڶ��ٿ�������������������Ԫ��������
| ʳ�� | ���ס����Źǡ����Ρ����⡢���������� |
| ���� | ʳ�Ρ����桢���ǡ�ʳ�ס�ζ�������͡�ʳ���� |
| ��Ʒ | �������������롢���ӡ����� |
��2��Ұ����У���һͬѧ���۷��������ˣ��۷䶾Һ�����ԣ�����ͬѧ�������÷���ˮΪ��ͿĨ���������֢״�����ᣬ������Ϊ����ˮ�ʼ��ԣ�
��3��Ұ��������С�ƴ������л�Ϥ��ijƷ�ƽ���ÿ100mL����Ӫ�����ı���������0.2g����Ӫ��������Ԫ����������Ϊ12.5%���ɴ˿ɼ���һƿ448mL��Ʒ�������������൱�ڶ��ٿ�������������������Ԫ��������
14��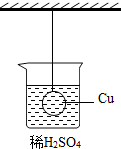 ��ͼ��ʾ��һ����ͭ���û��ϡ������Һ�У���ͭ�����ձ��Ӵ�������ϡ������Ͷ��һ��Ƭ������Ƭ��ַ�Ӧ��ͭ�����ܵ������仯����ǣ�������
��ͼ��ʾ��һ����ͭ���û��ϡ������Һ�У���ͭ�����ձ��Ӵ�������ϡ������Ͷ��һ��Ƭ������Ƭ��ַ�Ӧ��ͭ�����ܵ������仯����ǣ�������
 ��ͼ��ʾ��һ����ͭ���û��ϡ������Һ�У���ͭ�����ձ��Ӵ�������ϡ������Ͷ��һ��Ƭ������Ƭ��ַ�Ӧ��ͭ�����ܵ������仯����ǣ�������
��ͼ��ʾ��һ����ͭ���û��ϡ������Һ�У���ͭ�����ձ��Ӵ�������ϡ������Ͷ��һ��Ƭ������Ƭ��ַ�Ӧ��ͭ�����ܵ������仯����ǣ�������| A�� | ��� | B�� | ��С | C�� | ���� | D�� | ���ж� |
4������˵����ȷ���ǣ�������
| A�� | ����֧��ȼ�գ��������溽���е���Ҫȼ�� | |
| B�� | ��Ϊ�����뵪�����ܶȲ�ͬ�����Թ�ҵ�Ϸ���Һ̬��������ȡ���� | |
| C�� | ľ̿�ڿ�����ȼ�ղ��������о��ң���Ϊ���������������ϵ� | |
| D�� | �㡢Ϻ������ˮ�����棬����������������ˮ |
11��һ��̼ԭ�ӵ�����Ϊbǧ�ˣ������ԭ������Ϊ12��һ����ԭ�ӵ�����Ϊaǧ�ˣ������ԭ�ӵ����ԭ������Ϊ��������
| A�� | 16 | B�� | $\frac{b}{12a}$ | C�� | $\frac{a}{12b}$ | D�� | $\frac{12a}{b}$ |
8���������ֽ�����������ɫ�������ǣ�������
| A�� | �� | B�� | �� | C�� | �� | D�� | ͭ |