��Ŀ����
9��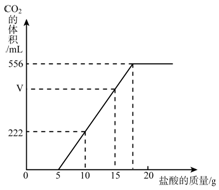 ����һ��Na2CO3��NaHCO3�Ļ���ijͬѧΪ�ⶨ�京����ȡһ�����ĸ���Ʒ����ˮ����μ���ϡ���Ტ���������ȷ�����Ӧ��Na2CO3+HCl=NaHCO3+NaCl��Ȼ��ŷ�����Ӧ��
����һ��Na2CO3��NaHCO3�Ļ���ijͬѧΪ�ⶨ�京����ȡһ�����ĸ���Ʒ����ˮ����μ���ϡ���Ტ���������ȷ�����Ӧ��Na2CO3+HCl=NaHCO3+NaCl��Ȼ��ŷ�����Ӧ��NaHCO3+HCl=NaCl+CO2��+H2O��
��֪�����¶���CO2�ܶ�Ϊ1.98g/L��ʵ���ò�����CO2�����ϡ����������������ͼ������㣺
��1��������������HC����������Ϊ7.3%
��2��ԭ��Ʒ��Na2CO3��NaHCO3���������Ƕ��٣�
���� ��1������ͼ����Կ���������5�����Ὺʼ������ų�������ͼ����Ϣ�г���ϵʽ���ɣ�
��2������ͼ���֪��5�������̼���Ʒ�Ӧ��д����ѧ����ʽ�����̼���ƺ�����̼�����Ƶ�������������̼����������556���������Լ�����ܵ�̼�����Ƶ�������
��� �⣺��1����ͼ����Կ���������5�������ʼ�ж�����̼������֣���ͼ����Կ��������루10-5��������ų�������̼�������222���������루15-5��������ų�������̼����������V����������V=222��2�T444������444 mLCO2������=$\frac{444mL}{1000mL/L}$��1.98g/L�T0.88�ˣ�
�����0.88gCO2ʱ������HCl������Ϊx��
NaHCO3+HCl=NaCl+CO2��+H2O
36.5 44
x 0.88g
$\frac{36.5}{44}$=$\frac{x}{0.88g}$
�ã�x=0.73g
��������Һ�����ʵ���������=$\frac{0.73g}{15g-5g}$��100%=7.3%��
��2�������5g����ʱ���μӷ�Ӧ��̼���Ƶ�����Ϊy������̼�����Ƶ�����Ϊz��
5�����������ʵ�����=5�ˡ�7.3%=0.365�ˣ�
Na2CO3+HCl�TNaHCO3+NaCl
106 36.5 84
y 0.365g z
$\frac{106}{y}$=$\frac{36.5}{0.365g}$=$\frac{84}{z}$
y=1.06g
z=0.84g
556mLCO2������=556mL��1000��1.98g/L=1.10�ˣ�
��ų�556mLCO2ʱ��Ӧ��NaHCO3����Ϊm��
NaHCO3+HCl=NaCl+CO2��+H2O
84 44
m 1.10g
$\frac{84}{44}$=$\frac{m}{1.1g}$
m=2.10g
��ԭ��Ʒ��NaHCO3������=2.1g-0.84g=1.26g��
�𣺢�������HCl����������Ϊ7.3%����ԭ��Ʒ����Na2CO31.06g��NaHCO3 1.26g��
���� ���ݻ�ѧ����ʽ����ʱ����һҪ��ȷ��д��ѧ����ʽ���ڶ�Ҫʹ����ȷ�����ݣ������������Ҫ������

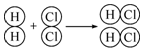
| A�� | �����������Է�������Ϊ32 | |
| B�� | ������ԭ�ӵ�ԭ�Ӻ���������Ϊ10 | |
| C�� | ������ԭ�ӵĺ��������Ϊ10 | |
| D�� | ������ԭ�Ӻ���ԭ�ӹ��ɵ�ˮ���ӣ�����Է�������Ϊ20 |
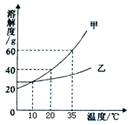
| A�� | ���ܽ�ȴ����ҵ��ܽ�� | |
| B�� | 35��ʱ100g����Һ�к�������60g | |
| C�� | 10��ʱ�ס���������Һ��������������һ����� | |
| D�� | �����¶ȿ�ʹ�ӽ����͵ļ���Һ��ɱ�����Һ |
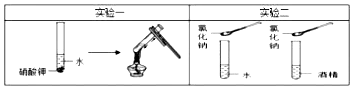
| A�� | ʵ��һĿ����̽���ܼ������ʶ�������ܽ��Ե�Ӱ�� | |
| B�� | ʵ��һ��Ҫ������ͬ�ı�����ˮ�����������ص����� | |
| C�� | ʵ�����̽���������ʶ������ܽ��Ե�Ӱ�� | |
| D�� | ʵ�������Ҫ������ͬ�ı�����ˮ�;ƾ������� |
| A�� | һ������ | B�� | �������� | C�� | ������̼ | D�� | PM2.5 |
| A�� | ������������ | B�� | ������2����ԭ�� | C�� | ��������Ԫ�� | D�� | ���������� |