��Ŀ����
11��ijũ����ѧ�Ļ�ѧ��ȤС���ڿ�չʵ��ʱ������ɽȪˮ��������������Һ����������������õ���Һ�����ֻ��ǵ�������һ��ʱ��������ײ������ְ�ɫ���������ȤС���ͬѧ��Դ����������չ������̽����������⣺��ɽȪˮ��������������ҺΪʲô����ֻ��ǣ���ɫ��������ʲô���ʣ�
�������룺������������Ʒ�����������������ʣ�
�������������ܽ���ɽȪˮ�е�ij���ʷ�����Ӧ����������ˮ�����ʣ�
��1��С��ͨ��ʵ��ܿ���ų��˲��������ʵ����ȡ����������Ʒ��������������ˮ��������ֻ��ǣ�
�������ϣ�����ɽȪˮ��Ӳˮ�����϶��̼����ƣ�̼�����[Ca��HCO3��2]���Ȼ�ֽ⣬Ҳ�����������Ʒ�Ӧ���Ҷ���̼������ɣ�
��2��С��ȡ��������ɽȪˮ���ձ��У������м��������ķ���ˮ�����裬�۲쵽��ĭ���٣����а�ɫ��״��ȣ�
������ȷ����ȡ��ɽȪˮ��Ӳˮ��
��3��С����С��ȡ����̼��Ƶij��������ʵ�飺ȡ�����ij��������������ϡ���ᣬʵ�������ǹ��岻���ܽ⣬����������ð������Ӧ�Ļ�ѧ����ʽ��CaCO3+2HCl=CaCl2+H2O+CO2����
��4����ɽȪˮ����������к���ˣ�����������ƣ����Ϳ�������������������Һ��
���� ��1������IΪ����������Կ���ѡ������ˮ��������֤��
��2������Ӳˮ�Ķ��弰����ˮ�����������
��3������̼��ƺ�ϡ���ᷴӦ�IJ������ж�ʵ������д���÷�Ӧ�Ļ�ѧ����ʽ��
��4���������⼰Ӳˮ�����ķ����������⣮
��� �⣺��1������I��˵���в��������ʣ����Կ��Խ��������Ƽ�������������ˮ�н����ܽ⣬���ȫ���ܽ⣬���Կ����ж�������������Ʒ�в������������ʣ�
��2��Ӳˮ�к��н϶�ĸ�þ���ӣ��������ˮ��������ĭ���٣�ͬʱ������϶�ĸ��������������ɽȪˮ�м������ˮ���裬�۲쵽���ٵ���ĭ�����жϸ�ɽȪˮΪӲˮ��
��3������ʵ����ۿ���֪���ó������к���̼��ƣ�����ʵ������Ӧ��Ϊ������٣�ͬʱ�������������ݣ��÷�Ӧ�Ļ�ѧ����ʽΪ��CaCO3+2HCl�TCaCl2+CO2��+H2O��
��4��ͨ�����������˵ķ���������Ӳˮ�����Ծ������������к���˵õ���ˮ������������������������Һ��
�ʴ�Ϊ��
��1��ȡ����������Ʒ��������������ˮ��������ֻ��ǣ�
��2����ĭ���٣����а�ɫ��״��ȣ���
��3�����岻���ܽ⣬����������ð����CaCO3+2HCl=CaCl2+H2O+CO2����
��4��������к���ˣ�..
���� ������Ҫ����ѧ��������ѧ��ѧ֪ʶ�ۺϷ����ͽ��ʵ�������������������ѧ�����������˼ά��ȣ�ǿ����ѧ������֪ʶ��������

 Ӧ�����������Ĵ���ѧ������ϵ�д�
Ӧ�����������Ĵ���ѧ������ϵ�д�| A�� | ֻ�Т� | B�� | ֻ�Т٢� | C�� | ֻ�Тڢۢ� | D�� | �٢ڢۢ� |
| A�� | K2MnO4��KMnO4��MnO2������MnԪ�صĻ��ϼ������� | |
| B�� | FeO��Fe2O3��Fe3O4 ���ߺ������������������� | |
| C�� | NH4NO3��NaCl��CaO��������ˮ��������Һ���¶������� | |
| D�� | NaCl��Na2CO3��NaOH���߱�����Һ��pH��� |
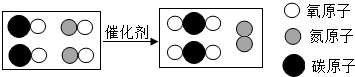 ͼΪ����β������װ���з�����Ӧ����ʾ��ͼ���й�����������ǣ�������
ͼΪ����β������װ���з�����Ӧ����ʾ��ͼ���й�����������ǣ�������| A�� | ͼ�е��ʵĻ�ѧʽΪN2 | B�� | �÷�Ӧ���û���Ӧ | ||
| C�� | ��Ӧ������������� | D�� | ��װ�ÿɾ�������β�� |
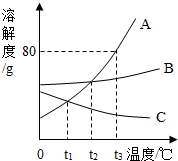
| A�� | һ���Ǽ� | B�� | �������� | C�� | ������������ | D�� | ���������� |
 ��ͼ��A��B��C���ֹ������ʵ��ܽ������ͼ������ͼʾ�ش��������⣺
��ͼ��A��B��C���ֹ������ʵ��ܽ������ͼ������ͼʾ�ش��������⣺ ��ͨ�ɵ���������е���;�ܹ㣬�乹��ʾ��ͼ��ͼ���ش��������⣺
��ͨ�ɵ���������е���;�ܹ㣬�乹��ʾ��ͼ��ͼ���ش��������⣺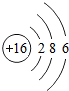 ����ԭ�ӵ�������Ϊ16����ԭ����3�����Ӳ㣻��ԭ���ڻ�ѧ�仯���ã���á���ʧ�������ӣ���Ԫ�����ڷǽ�������������ǽ�������Ԫ�أ�
����ԭ�ӵ�������Ϊ16����ԭ����3�����Ӳ㣻��ԭ���ڻ�ѧ�仯���ã���á���ʧ�������ӣ���Ԫ�����ڷǽ�������������ǽ�������Ԫ�أ�