��Ŀ����
��7�֣�������������������̼��һ����̼�dz��л�ѧ�еij������ʣ���ͼ�����ǵ�һЩ����Ӧ�á�
| A��̼������ | B����¯���� | C���ɱ� | D��ȼ��ȼ�� |
��1������������������˹�����������ǣ����������ƻ�ѧʽ����ͬ�� ��
��2��������������̼�����ϵ������� �����з����Ļ�ѧ����ʽΪ ��
��3����¯������һ����̼��ԭ�������Ļ�ѧ����ʽΪ ��
��4��������ȼ�ϵ������� ����ȼ�յĻ�ѧ����ʽΪ ��
��1���ɱ�����CO2�� ����������������������������������������1��
��2��CO2 CO2 + H2O ="=" H2CO3 ������������������������������������2��
��3��3CO + Fe2O3 ="===" 2Fe + 3CO2 (���¡����ȶ�����)����������������2��
��4��CO��H2��ȱһ�����֣�
2CO + O2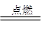 2CO2 2H2 + O2
2CO2 2H2 + O2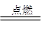 2H2O ����������������2��
2H2O ����������������2��
����

��ϰ��ϵ�д�
 �Ͻ�ƽСѧ��������ϵ�д�
�Ͻ�ƽСѧ��������ϵ�д� �Ƹ������������ϵ�д�
�Ƹ������������ϵ�д�
�����Ŀ
���и������ʣ�ֻ��ˮ���ܼ���������ǣ�������
| A��ʳ�ι��塢̼��ƹ��塢������ع��� | B��������������������̼ | C�����͡��ƾ���Ũ���� | D���������ƹ��塢�Ȼ��ƹ��塢����粒��� |