��Ŀ����
�������ϴ��е����þ���ϩ [ (CH2CH2)n ] �Ƴɣ��е����þ�����ϩ [ (CH2CHCl)n ] �Ƴɣ�ͨ���������Ͽ�֪��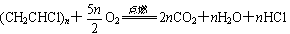 ������n����ǧ���������ֵ��HCl���弫������ˮ���д̱���ζ����ˮ��Һ���������������ķ����Ǽ���AgNO3��Һ���۲��Ƿ������ɫ������
������n����ǧ���������ֵ��HCl���弫������ˮ���д̱���ζ����ˮ��Һ���������������ķ����Ǽ���AgNO3��Һ���۲��Ƿ������ɫ������
�� ͨ����ȼ�ķ������Լ������ϩ�;�����ϩ��������ϴ���ȼʱ��ǿ�ҵĴ̱���ζ���������ϴ��������� �Ƴɵġ�
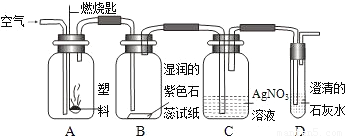
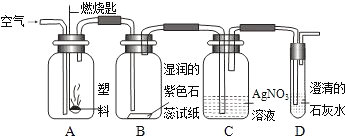
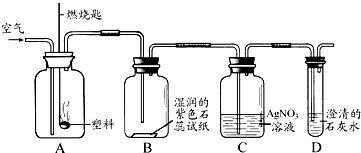
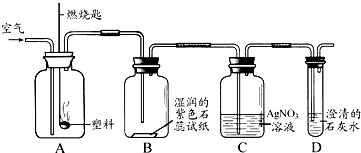
�� ijͬѧ���������ͼ��ʾʵ����̽������ȼ�յIJ��
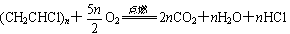 ������n����ǧ���������ֵ��HCl���弫������ˮ���д̱���ζ����ˮ��Һ���������������ķ����Ǽ���AgNO3��Һ���۲��Ƿ������ɫ������
������n����ǧ���������ֵ��HCl���弫������ˮ���д̱���ζ����ˮ��Һ���������������ķ����Ǽ���AgNO3��Һ���۲��Ƿ������ɫ�������� ͨ����ȼ�ķ������Լ������ϩ�;�����ϩ��������ϴ���ȼʱ��ǿ�ҵĴ̱���ζ���������ϴ��������� �Ƴɵġ�
�� ijͬѧ���������ͼ��ʾʵ����̽������ȼ�յIJ��
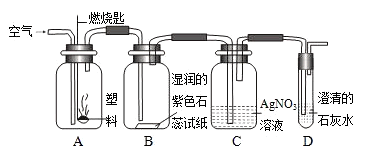
��Aƿȼ�յ��Ǿ�����ϩ���ϣ���Bƿ�п��Կ����������� ��C��D��ƿ�з�����Ӧ�Ļ�ѧ����ʽΪ �� ����Aƿ���õ��Ǿ���ϩ���ϣ�����Cƿ���ܹ۲쵽�������� ��
�� ������ϩ
�� ��ֽ��죬ƿ�ڱ���ˮ������ƿ���а�������������������
HCl+AgNO3=== AgCl��+HNO3
CO2+Ca(OH)2===CaCO3��+H2O
��Һ��û�а�ɫ������������ð��
�� ��ֽ��죬ƿ�ڱ���ˮ������ƿ���а�������������������
HCl+AgNO3=== AgCl��+HNO3
CO2+Ca(OH)2===CaCO3��+H2O
��Һ��û�а�ɫ������������ð��

��ϰ��ϵ�д�
 ����������ϵ�д�
����������ϵ�д� �Ż���ҵ�Ϻ��Ƽ����׳�����ϵ�д�
�Ż���ҵ�Ϻ��Ƽ����׳�����ϵ�д�
�����Ŀ
 O2
O2  2nCO2+nH2O+nHCl
2nCO2+nH2O+nHCl
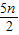 O2
O2 2nCO2+nH2O+nHCl������n����ǧ���������ֵ��HCl���弫������ˮ���д̱���ζ����ˮ��Һ���������������ķ����Ǽ���AgNO3��Һ���۲��Ƿ������ɫ������
2nCO2+nH2O+nHCl������n����ǧ���������ֵ��HCl���弫������ˮ���д̱���ζ����ˮ��Һ���������������ķ����Ǽ���AgNO3��Һ���۲��Ƿ������ɫ������