��Ŀ����
ʵ���ҳ�������װ���о��������ȡ�����ʣ�������ѧ֪ʶ�ش��������⡣
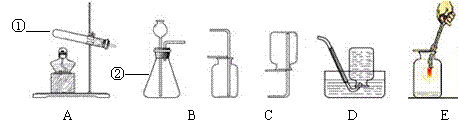 |
�� д�����������Ƣ�________________ ��___________________
�� д��ʵ������ȡ������һ����ѧ����ʽ________________________________������
�÷�Ӧ��ȡ����������װ��Ӧѡ__________������ţ���ͬ����
�� һ����������������ˮ���ڿ��������������з�Ӧ2NO+O2==2NO2�����ռ�һ�����������װ���ǣ�����ţ�___________��
�� ��ͼE��ϸ��˿��������ȼ�յ�ʵ�飬�ҹ۲쵽��������_____________________
_________________________������صĻ�ѧ����ʽ��__________________________��
�� ʵ�����ォ�ƵõĶ�����̼����ͨ�����ʯ��ˮ�У�ʼ��δ�������������ܵ�ԭ����__________ ____________________________________��
____________________________________��
�� �Թ�, ��ƿ ��2KMnO4 === K2MnO4 + MnO2 +O2��;
|
��2KClO3 ===== 2KCl + 3O2��; 2H2O2 ==== 2H2O + O2������A
(ע���� ���ǹ�����������������װ��ѡ��B)
���ǹ�����������������װ��ѡ��B)
|
3Fe + 2O2 ==== Fe3O4 ������д��һ�����ɣ�
�� װ�������Բ��ã������Ķ�����̼ɢ�ݵ��������˻�������лӷ��ԣ��ӷ��������Ȼ����������ʯ��ˮ���γ����ᣬ�ܽ������ɵ�̼��Ƴ������𰸺������÷֣�

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�

 ��ͼ��ʵ���ҳ��õ�ʵ����������ش��������⣺
��ͼ��ʵ���ҳ��õ�ʵ����������ش��������⣺