��Ŀ����
���պ����ʵ����ʡ���;�ͻ�ѧ������ѧ�û�ѧ�Ļ������������ѡ�����գ�
��1���Ҵ�¶���ڿ������������٣�����Ϊ���� ��
A����ԭ�� B����ˮ�� C���ӷ���
��2��ʯī�����缫������������ ��
A�������� B�������� C����ȼ��
��3�����Ƹ��ʱ�������ͷ۵��� ��
A��С�մ� B�������� C����ʯ��
��4��������Ȼ�߷��Ӳ��ϵ��� ��
A�������� B������ϩ C����ë
��5���ú��ʵķ��ű�ʾ���������и�Ԫ�صĻ��ϼ�Ϊ+2 ��
��6����1��7��8��Ԫ���γɵ�һ���εĻ�ѧʽ�ֱ�Ϊ ��
��1���Ҵ�¶���ڿ������������٣�����Ϊ����
A����ԭ�� B����ˮ�� C���ӷ���
��2��ʯī�����缫������������
A�������� B�������� C����ȼ��
��3�����Ƹ��ʱ�������ͷ۵���
A��С�մ� B�������� C����ʯ��
��4��������Ȼ�߷��Ӳ��ϵ���
A�������� B������ϩ C����ë
��5���ú��ʵķ��ű�ʾ���������и�Ԫ�صĻ��ϼ�Ϊ+2
��6����1��7��8��Ԫ���γɵ�һ���εĻ�ѧʽ�ֱ�Ϊ
���㣺���顢�Ҵ��ȳ����л�������ʺ���;,�����ε���;,̼���ʵ��������ʼ���;,��ѧʽ����д������,��ѧ���ż�����Χ���ֵ�����,�л��߷��Ӳ��ϵķ��༰����
ר�⣺���ʵ���������;,��ѧ����������غ㶨��
��������1���Ҵ����лӷ��ԣ�
��2��ʯī���е����ԣ�
��3��̼�����Ƽ����ֽ⣻
��4�����ݲ��ϵķ�����������
��5������Ԫ�ػ��ϼ۵ı�ʾ��������д��
��6��ԭ������������Ԫ�ص����࣬�����εĻ�ѧʽ��д�������з�����
��2��ʯī���е����ԣ�
��3��̼�����Ƽ����ֽ⣻
��4�����ݲ��ϵķ�����������
��5������Ԫ�ػ��ϼ۵ı�ʾ��������д��
��6��ԭ������������Ԫ�ص����࣬�����εĻ�ѧʽ��д�������з�����
����⣺��1���Ҵ��ӷ������Գ��ڷ����ڿ�������������٣����C��
��2��ʯī�����Ƶ缫������Ϊʯī���е����ԣ����B��
��3��С�մ���̼�����Ƶ��׳ƣ��ڼ��ȵ������·ֽ�Ϊ̼���ơ�ˮ�Ͷ�����̼�����Ƿ��ͷ۵���Ҫ�ɷ֣����A��
��4�������Dz��Ǹ߷��ӻ��������ϩ���л��ϳɸ߷��Ӳ��ϣ���ë������Ȼ�߷��Ӳ��ϣ����C��
��5����עԪ�ػ�ԭ���ŵĻ��ϼ�ʱ��Ҫд��Ԫ�ط��Ż�ԭ���ŵ����Ϸ����ǡ�1��ʱ����ʡ�ԣ�������������ǰ����ֵ�ں��������и�Ԫ�صĻ��ϼ�Ϊ+2������������и�Ԫ�صĻ��ϼ۱�ʾΪ��
O�����
O��
��6��1��7��8��Ԫ�طֱ�Ϊ��Ԫ�ء���Ԫ�غ���Ԫ�أ���ɵ���������泥����NH4NO3��
��2��ʯī�����Ƶ缫������Ϊʯī���е����ԣ����B��
��3��С�մ���̼�����Ƶ��׳ƣ��ڼ��ȵ������·ֽ�Ϊ̼���ơ�ˮ�Ͷ�����̼�����Ƿ��ͷ۵���Ҫ�ɷ֣����A��
��4�������Dz��Ǹ߷��ӻ��������ϩ���л��ϳɸ߷��Ӳ��ϣ���ë������Ȼ�߷��Ӳ��ϣ����C��
��5����עԪ�ػ�ԭ���ŵĻ��ϼ�ʱ��Ҫд��Ԫ�ط��Ż�ԭ���ŵ����Ϸ����ǡ�1��ʱ����ʡ�ԣ�������������ǰ����ֵ�ں��������и�Ԫ�صĻ��ϼ�Ϊ+2������������и�Ԫ�صĻ��ϼ۱�ʾΪ��
| +2 |
| Ca |
| +2 |
| Ca |
��6��1��7��8��Ԫ�طֱ�Ϊ��Ԫ�ء���Ԫ�غ���Ԫ�أ���ɵ���������泥����NH4NO3��
�����������ѶȲ������ʵ����ʾ������ʵ���;�����ճ������ʵ���������;��������ѧ�����ѧʽ�����ӷ��ŵȣ�����д������ȷ����⣮

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
ϴ�·۵���Ч�ɷ��Ǵ������[Ca��ClO��2]��������Ԫ�صĻ��ϼ�Ϊ��������
| A��+2 | B��+1 | C��-1 | D��+5 |
 H7N9�������������ĸ߶ȹ�ע��2013��4��5�գ�һ�����͵Ŀ�������ҩ������Τ�Ȼ���ע��Һ������ʳƷҩƷ�ල�����ּܾ��������У�������Τ�Ļ�ѧʽΪC15H28N4O4�����ж�������Τ���й�˵����ȷ���ǣ�������
H7N9�������������ĸ߶ȹ�ע��2013��4��5�գ�һ�����͵Ŀ�������ҩ������Τ�Ȼ���ע��Һ������ʳƷҩƷ�ල�����ּܾ��������У�������Τ�Ļ�ѧʽΪC15H28N4O4�����ж�������Τ���й�˵����ȷ���ǣ�������| A�����������ɶ���ԭ�ӹ��ɵĻ����� |
| B���������к���28����ԭ�� |
| C����������̼���⡢����������Ԫ����� |
| D����������̼����Ԫ�ص�������Ϊ15��28 |
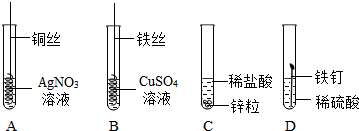
 ����ͬѧ����һ����Ȥ��ʵ�飬װ����ͼ��
����ͬѧ����һ����Ȥ��ʵ�飬װ����ͼ�� ú��ʯ�ͺ���Ȼ���Ȼ�ʯȼ����Ŀǰ����ʹ�õ�����Ҫȼ�ϣ�Ҳ����Ҫ�Ļ���ԭ�ϣ�
ú��ʯ�ͺ���Ȼ���Ȼ�ʯȼ����Ŀǰ����ʹ�õ�����Ҫȼ�ϣ�Ҳ����Ҫ�Ļ���ԭ�ϣ�