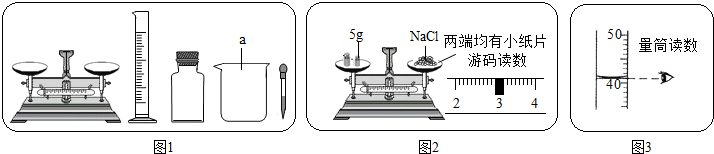
��Ŀ����
ijʵ��С������ͼ1��ʾ�������С�����һ�������������Ȼ�����Һ����ʵ�飺
��1����ɴ�ʵ����ȷ�IJ���˳���� ������ţ���
���ܽ� �ڰ���õ���Һװ���Լ�ƿ �۳��� �ܼ���
��2����ʵ��Ҫ��ͼ1�л�ȱ�ٵIJ��������� �������ƣ����Ȼ��ƹ�����ˮ�е��ܽ��ٶȽ�������д��һ�ּӿ��Ȼ��ƹ����ܽ��ٶȵĴ�ʩ�� ���������a��������ˮ����ֱ�ӵ����Ȼ��ƹ�������ܽ⣬�������Ƶ��Ȼ�����Һ���������� ���ƫ����ƫС������Ӱ�족����
��3��Сԭ��ͼ2�IJ�����ȡ�Ȼ��ƣ�С����Сԭ�IJ����д�������ΪСԭ�����еĴ����� ��
��4��С��ȡ���������Ƶ���Һ���Թ��У������е���2��ʯ����Һ������Һ����ɫΪ ��
���𰸡���������1������������Һ�IJ���ش�
��2��������Һ��Ҫ�IJ����������ձ�����Ͳ���ιܡ����������ӿ��Ȼ��ƹ����ܽ��ٶȿ��Խ��衢���Ȼ�ı��������С���������ʵ�������������������
��3��������ƽ�������ʵ�����ʱӦ�������룻
��4��ʯ����Һ��������Һ�в���ɫ��
����⣺��1��������Һ�IJ����Ǽ��㡢�������ܽ⡢װƿ��
��2��������Һ��Ҫ�IJ����������ձ�����Ͳ���ιܡ����������ӿ��Ȼ��ƹ����ܽ��ٶȿ��Խ��衢���Ȼ��С������С�����ʵ���������= ×100%������������ȷ���ܼ����������ӣ�����Һ���������ӣ������ʵ���������ƫС��
×100%������������ȷ���ܼ����������ӣ�����Һ���������ӣ������ʵ���������ƫС��
��3����ƽ�������ʵ�����ʱӦ�������룬ͼ�зŷ��ˣ�
��4��ʯ����Һ��������Һ�в���ɫ���Ȼ�����Һ�����ԣ�
�ʴ�Ϊ��
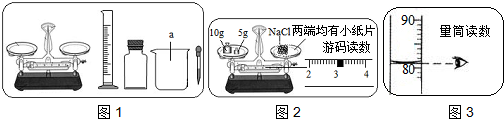
��1���ܢۢ٢ڣ�
��2���������� ���衢���Ȼ��С������С�� ƫС��
��3������ʱ������Ȼ��Ƶ�λ�÷ŷ��ˣ�
��4����ɫ��
���������⿼��������һ��������������Һ�IJ��衢������������������֪ʶ���ѶȲ���ϸ�ķ������ɣ�
��2��������Һ��Ҫ�IJ����������ձ�����Ͳ���ιܡ����������ӿ��Ȼ��ƹ����ܽ��ٶȿ��Խ��衢���Ȼ�ı��������С���������ʵ�������������������
��3��������ƽ�������ʵ�����ʱӦ�������룻
��4��ʯ����Һ��������Һ�в���ɫ��
����⣺��1��������Һ�IJ����Ǽ��㡢�������ܽ⡢װƿ��
��2��������Һ��Ҫ�IJ����������ձ�����Ͳ���ιܡ����������ӿ��Ȼ��ƹ����ܽ��ٶȿ��Խ��衢���Ȼ��С������С�����ʵ���������=
 ×100%������������ȷ���ܼ����������ӣ�����Һ���������ӣ������ʵ���������ƫС��
×100%������������ȷ���ܼ����������ӣ�����Һ���������ӣ������ʵ���������ƫС����3����ƽ�������ʵ�����ʱӦ�������룬ͼ�зŷ��ˣ�
��4��ʯ����Һ��������Һ�в���ɫ���Ȼ�����Һ�����ԣ�
�ʴ�Ϊ��
��1���ܢۢ٢ڣ�
��2���������� ���衢���Ȼ��С������С�� ƫС��
��3������ʱ������Ȼ��Ƶ�λ�÷ŷ��ˣ�
��4����ɫ��
���������⿼��������һ��������������Һ�IJ��衢������������������֪ʶ���ѶȲ���ϸ�ķ������ɣ�

��ϰ��ϵ�д�
�����Ŀ