��Ŀ����
11�� ��֪Na2CO3��ˮ��Һ�ʼ��ԣ���һ�ձ���ʢ��Na2CO3��NaCl��ɵĹ�������20.4g���������μ������ʷ���Ϊ10%��ϡ���ᣬ�ų��������������������ϡ�����������ϵ��ͼ��ʾ�����������ش����⣺
��֪Na2CO3��ˮ��Һ�ʼ��ԣ���һ�ձ���ʢ��Na2CO3��NaCl��ɵĹ�������20.4g���������μ������ʷ���Ϊ10%��ϡ���ᣬ�ų��������������������ϡ�����������ϵ��ͼ��ʾ�����������ش����⣺��1�����μ�ϡ������ͼ��B��ʱ���ձ�����Һ��pH��7���������
��=��������������
��2�����μ�ϡ������ͼ��A��ʱ���ձ���Ϊ��������Һ�����£��������������̼���Ƶ�������
���� ����ͼʾ���з��������μ�ϡ���ᵽA��ʱ��̼������ȫ��Ӧ����Ӧ���������Ȼ��ƣ�ԭ���Ĺ���������Ҳ���Ȼ��ƣ����������Ȼ����������������������е�̼���Ƶ�������
��� �⣺
��1����ͼ��֪�����μ�ϡ������A��ʱ��������ȫ��Ӧ�����Ե��μӵ�B��ʱ������������ձ�����Һ��PH��7��
��2����μ�ϡ���ᵽAʱ���μӷ�Ӧ��̼���Ƶ�����Ϊx��
�������֪�����μӵ�Aʱ�������Ȼ����������73g��10%�T7.3g
Na2CO3+2HCl=2NaCl+H2O+CO2��
106 73
x 7.3g
$\frac{106}{x}=\frac{73}{7.3g}$
x=10.6g
�𰸣�
��1������
��2������������̼���Ƶ�����Ϊ10.6�ˣ�
���� �����ڼ���ʱ����Һ�����ļ����ǹؼ������׳�����Ӧ���������غ㣬��Ӧ����Һ������=����������+����ϡ��������-�ų�����������

��ϰ��ϵ�д�
 ��ѧȫ��������ѵ��ϵ�д�
��ѧȫ��������ѵ��ϵ�д�
�����Ŀ
1��ijѧϰС���������������о���
��1������ϡ����������²�������Һ��pH���ߵ���ABC������ĸ����
A��������ˮ B������������
C���������������� D��������Ũ����
��2���������ձ��зֱ�ע���������ᣬ�ף�������������Ϊ98%���ܶ�Ϊ1.84g/cm3�����ң��������ˮ�İ������1��1��ϣ���10ml����������¼��ͬʱ�䣨h������������Һ��ˮ�����������ݼ�����
������ȡ�����ʱ���Ӷ�����������������ȷ������£����������ҵ���������������ƫС���ƫ����ƫС�������ܡ�����
���ɱ���ʵ�����ݿ�֪���й�������ˮ�Ե�˵���������B������ĸ����
A��������Ҷ�����ˮ�� B��ֻ�����������ˮ�� C���������ˮ�Խ�������ǿ
��3��С��ͬѧ����ϡ�ͺ��ϡ��������ᱵ���������ơ�̼��ص����ʵ���Һ�ԡ����ֽⷴӦ������������������̽����
�ٽ�����������Һ������ϣ��ж�������֮���ܷ�����Ӧ����д�����з����кͷ�Ӧ�Ļ�ѧ����ʽH2SO4+2NaOH�TNa2SO4+2H2O��
�����������������ʵ���Һ���ʵ����������ʻ�ϲ����ˣ��õ�һ����ɫ��Һ�����ⶨ������Һ�����ԣ����ڸ���Һ��һ�����е�������H+��Na+��K+��NO3-�����ܺ��е�������Ba2+��SO42-��
��1������ϡ����������²�������Һ��pH���ߵ���ABC������ĸ����
A��������ˮ B������������
C���������������� D��������Ũ����
��2���������ձ��зֱ�ע���������ᣬ�ף�������������Ϊ98%���ܶ�Ϊ1.84g/cm3�����ң��������ˮ�İ������1��1��ϣ���10ml����������¼��ͬʱ�䣨h������������Һ��ˮ�����������ݼ�����
| ʱ��/h | 1 | 1.5 | 4 | 8 | 12 | 24 | 48 | 60 | |
| ��ˮ����/g | ����� | 1.6 | 2.2 | 5.2 | 10.3 | 14.0 | 20.9 | 29.2 | 32.1 |
| ������ | 1.2 | 1.5 | 3.5 | 5.9 | 8.1 | 12.9 | 19.5 | 21.0 | |
���ɱ���ʵ�����ݿ�֪���й�������ˮ�Ե�˵���������B������ĸ����
A��������Ҷ�����ˮ�� B��ֻ�����������ˮ�� C���������ˮ�Խ�������ǿ
��3��С��ͬѧ����ϡ�ͺ��ϡ��������ᱵ���������ơ�̼��ص����ʵ���Һ�ԡ����ֽⷴӦ������������������̽����
�ٽ�����������Һ������ϣ��ж�������֮���ܷ�����Ӧ����д�����з����кͷ�Ӧ�Ļ�ѧ����ʽH2SO4+2NaOH�TNa2SO4+2H2O��
�����������������ʵ���Һ���ʵ����������ʻ�ϲ����ˣ��õ�һ����ɫ��Һ�����ⶨ������Һ�����ԣ����ڸ���Һ��һ�����е�������H+��Na+��K+��NO3-�����ܺ��е�������Ba2+��SO42-��
6����ѧ�����ʹ���ѧϰ��ѧ������������ȷ������й�������Ϊ��ȷ���ǣ�������
| A�� | SO2��SO3�ķ��ӽṹ��ͬ�����Ի�ѧ���ʲ�ͬ | |
| B�� | ͨ�����ʳ��ˮ�ķ��������Ƶ�NO2 | |
| C�� | ������Ʒ����Ȼ�������κλ�ѧ���� | |
| D�� | ���ü�ȩ��Һ���ݸ�����Ʒ���� |
16��С��ͬѧ��ij�������ʽ�������ͼ��ʾ��ʵ�飬����ʵ���ж�����˵������ȷ���ǣ�������


| A�� | �ù������ʵ��ܽ�����¶ȵ����߶����� | |
| B�� | ��Һbһ���Dz�������Һ | |
| C�� | ��Һa����������������Һb�е��������� | |
| D�� | ��Һb��������������С����Һc�������������� |
3�������ơ��ǻ�ѧѧϰ�����г��õ�˼ά�����������������ƽ����������ȷ���ǣ�������
| A�� | ��Ϊ����Һ�ʼ��ԣ����Գʼ��Ե���Һһ���Ǽ���Һ | |
| B�� | �����������ᷴӦ����AlCl3��H2�����Խ����������ᷴӦ����FeCl3��H2 | |
| C�� | ̼�ĵ��ʽ��ʯ��ʯī������̼ԭ�ӹ��ɣ���������̼����Ҳ����̼ԭ�ӹ��� | |
| D�� | ��ȼ����ǰ��Ҫ���鴿�ȣ����Ե�ȼ����ǰҲ��Ҫ���鴿�� |

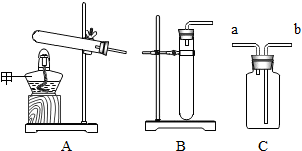 ʵ����������ͼʵ��װ�ý����йػ�ѧʵ�飬�ش��������⣺
ʵ����������ͼʵ��װ�ý����йػ�ѧʵ�飬�ش��������⣺