��Ŀ����
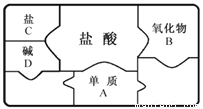
����ͼ���ܷ�ӳ����ʵ����( )
A. 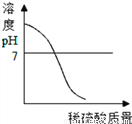 ��NaOH��Һ����μ���ϡ����
��NaOH��Һ����μ���ϡ����
B. 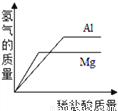 ������������þ�м�������ϡ����
������������þ�м�������ϡ����
C. 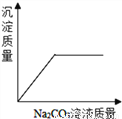 ��ϡHCl��CaCl2�Ļ����Һ����μ���Na2CO3��Һ
��ϡHCl��CaCl2�Ļ����Һ����μ���Na2CO3��Һ
D. 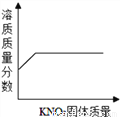 �������������ӽ����͵��������Һ�в��ϼ�������ع���
�������������ӽ����͵��������Һ�в��ϼ�������ع���
��ϰ��ϵ�д�
�����Ŀ
Ϊ�Ƚ���ͬŨ�ȵ�FeCl3��Һ��CuSO4��Һ��H2O2�ֽ�Ĵ�Ч����ij�о�С�����������̽����
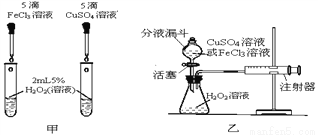
(1)��д��H2O2�ֽ�Ļ�ѧ����ʽ_______________��
�������о���(2)��ͼ�ף���ͨ���۲�__________�����ԱȽ����ߵĴ�Ч����
�������о���(3)Ҫ�����Ƚ����ߵĴ�Ч�����ɲ������ɵ�������������____________��
�������о���(4)��CuSO4��Һ�У�����������������������أ�
����1��ͭ���ӣ�Cu2+��������ã�
����2��___________������ã�
����3��___________������ã�
��ע�⣺����Եڣ�5��С�⽱��4�֣���ѧ�Ծ��ֲܷ�����60�֡���
��5�������ʵ�飬��֤����1
ʵ����� | ʵ�������� |
_______________ | ______________ |


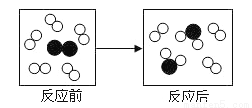

 ______________________
______________________ _____________________
_____________________