��Ŀ����
�����벻����ѧ��
��ʳ����Լ����3%-5%�Ĵ��ᣨC2H4O2����������_________��Ԫ����ɡ�C2H4O2��Ħ������Ϊ_________��0.5mol C2H4O2������Լ����_________����ԭ�ӡ�
����ͼ�ǵ��ˮ��װ�ã�д��ˮ���Ļ�ѧ����ʽ__________________����������ķ��Ӹ�������_______��������������ʵ�������֮����ȡ����³�ѹ�£����������������ܽ�ȷֱ�Ϊ1.63��10-3g/100gˮ��4.34��10-3g/100gˮ���ڵ������У��ס����������������ȿ���________������ڡ���С�ڡ�����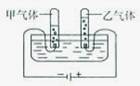
3��60g/mol��6.02��1023��2H2O 2H2��+O2�������ʵ���������
2H2��+O2�������ʵ���������
����

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
�����벻����ѧ����������������ǣ�������
| A���ó涣ҧʱ���˵�Ƥ���ڷ��ڳ����ᣬ�ڶ�ҧ��ͿĨ����ˮ����ʹ�� | B����ʳ��ˮ����ˮƿ���ڱ��ϳ�����ˮ�� | C����ͥ�������ˮ�ķ�������ˮ��Ӳ�� | D�������������⣬����ʳ��ȥ�� |
 �����벻����ѧ����ѧ������ϢϢ��أ�
�����벻����ѧ����ѧ������ϢϢ��أ�