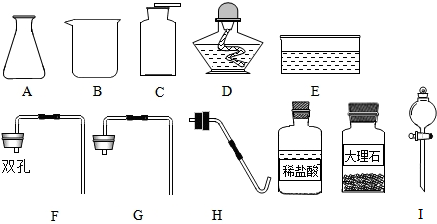
��Ŀ����
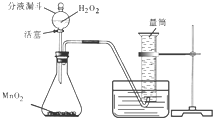 Ϊ�ռ�30mL������С����չ������ʵ�飺
Ϊ�ռ�30mL������С����չ������ʵ�飺���������������װ�������ԣ�
��ȡ�����Ķ������̷�ĩ������ƿ�У�ȡһ��������������Һ�������Һ©���У�
����ͼ��ʾ��װ��ʵ��װ�ã�
������Һ©����������ȡ���ռ�������
����������ƿ�еĶ������̣�ϴ�������Ż�ԭ����
�ش��������⣺
��1���÷�Ӧ�Ļ�ѧ����ʽΪ
��2����ʵ���У��ռ������ķ�����
��3���ռ�����ʱ��������Ӷ��������ռ�����ʵ�����
��4���������̿��Ի��������õ�ԭ����
���㣺��������ȡװ��,��������-��Ͳ,�������ռ�����,��д��ѧ����ʽ�����ֱ���ʽ�����뷽��ʽ
ר�⣺���������ʵ�����Ʒ������顢�����뾻��
��������1��ͨ������£����������ڶ������̵Ĵ������£��ֽ�����ˮ��������
��2���ռ�����ķ��������ſ���������ˮ����
��3���������������ʱ��������Ӷ�����������Ľ��ƫС��
��4�������ڷ�Ӧǰ��������ͻ�ѧ���ʲ��䣮
��2���ռ�����ķ��������ſ���������ˮ����
��3���������������ʱ��������Ӷ�����������Ľ��ƫС��
��4�������ڷ�Ӧǰ��������ͻ�ѧ���ʲ��䣮
����⣺
��1�����ø�װ����ȡ��������Ҫ���ȣ�Ӧ�������ù���������ȡ���������������ڶ������̵Ĵ������·ֽ�Ļ�ѧ����ʽΪ��2H2O2
2H2O+O2����
���2H2O2
2H2O+O2����
��2����ʵ���У��ռ������ķ�������ˮ��������
�����ˮ��������
��3���������������ʱ��������Ӷ�������������С��ʵ��ֵ��
���С�ڣ�
��4���������̿��Ի��������õ�ԭ���������ͻ�ѧ���ʲ��䣮
��������ͻ�ѧ���ʲ��䣮
�𰸣�
��1��
��2����ˮ������
��3����
��4�������ͻ�ѧ���ʶ����ı�
��1�����ø�װ����ȡ��������Ҫ���ȣ�Ӧ�������ù���������ȡ���������������ڶ������̵Ĵ������·ֽ�Ļ�ѧ����ʽΪ��2H2O2
| ||
���2H2O2
| ||
��2����ʵ���У��ռ������ķ�������ˮ��������
�����ˮ��������
��3���������������ʱ��������Ӷ�������������С��ʵ��ֵ��
���С�ڣ�
��4���������̿��Ի��������õ�ԭ���������ͻ�ѧ���ʲ��䣮
��������ͻ�ѧ���ʲ��䣮
�𰸣�
��1��
��2����ˮ������
��3����
��4�������ͻ�ѧ���ʶ����ı�
�������������ʵ�飬��ѧ�ؽ���ʵ�顢����ʵ�飬�ǵó���ȷʵ����۵�ǰ�ᣬ���Ҫѧ�����ʵ�顢����ʵ�顢����ʵ�飬Ϊѧ�û�ѧ֪ʶ�춨������

��ϰ��ϵ�д�
�����Ŀ
���б仯������ֻ���������仯���ǣ�������
| A����ȼ�շ�������ë�ͺϳ���ά |
| B��������������౬ը |
| C��Ϊ�Ը���Ϊԭ����� |
| D��������Ʒ���� |
����ʵ�������ȷ���ǣ�������
A�� ��ȼ�ƾ��� |
B�� ��֤������̼���� |
C��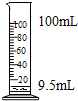 ��ȡҺ�� |
D�� �����Ȼ�����Һ |