��Ŀ����
����������������ͬ�������Ϲ�ϵ���У�
��һ�������Ĺ㷺Ӧ��
��1����������ͨ����Ϊ�����֣����������Ͻ����Ҫ���������_________��д����������ɸ֡��з�����Ӧ�Ļ�ѧ����ʽ_________��
��2��������������;�ˮ������ѧԭ���ǣ�4K2FeO4+10H2O=4X+3O2+8KOH��X�Ļ�ѧʽ��_________ ��
��������������ʴ������
��1���������������Ʒ����������� _________������ţ���
a. ��ʪ������ b. ��������� c. ���ֽ���ʳ��ˮ��
��2���������д�������ת����Fe(OH)2 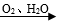 Fe(OH)3���÷�Ӧ�Ļ�ѧ����ʽΪ_________��
Fe(OH)3���÷�Ӧ�Ļ�ѧ����ʽΪ_________��
������������ɷ����ⶨ
��1��������һ����Fe2O3•nH2O�����ܺ�FeCO3��FeCO3��ϡ����ķ�Ӧ��CaCO3����)������ij������Ʒ�м�ϡ���ᣬ_________��������֤������FeCO3��
��2����ȡ23.2gֻ��Fe2O3•nH2O������������Ʒ������ͼװ��ʵ�飮
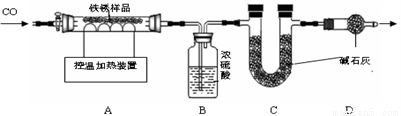
���������ϡ�
��Ũ���������ˮ�ԣ���ʯ��������ˮ�Ͷ�����̼���壮
����110��ʱ��Fe2O3•nH2O��ȫ�ֽ�ΪFe2O3��H2O��
��500��ʱFe2O3�ſ�ʼ����ԭ�����¶Ȳ�ͬʱ���������Fe3O4��FeO����ɫ����Fe��
��3����װ���м���������Ʒǰ������еIJ�����________��
��4��ʵ��ʱ����ͨ��CO��Ŀ���� _________ ��
��5�������ȳ���500��ʱ���۲쵽A�е�������_________��
��6���±�Ϊ������700��ʱ�������ݣ���ͼΪA�й��������ͼ����¶ȹ�ϵͼ

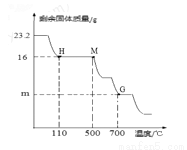
�ش���������
��Fe2O3•nH2O��n��ֵΪ_________��ͼ��m��ֵΪ_________��
������װ��D��������n��ֵ_________���ƫ����ƫС�����䡱��
��700������ʱ�����ڹ������Ļ�ѧʽΪ_________��
�ܸ�װ����һ����ȱ�ݣ���θĽ���_________��
����ʵ������У�������Ϊ���ʣ����ܴﵽʵ��Ŀ�ĵ���
ѡ�� | ���� | Ŀ�� | ��Ҫʵ����� |
A | CO��H2 | ���� | ��ȼ�������Ϸ��ָ����ձ����۲����� |
B | KNO3��NaCl�� | �ᴿ | ��90��ˮ�ܽ⣬���½ᾧ������ |
C | H2��HCl�� | ���� | ͨ��װ��̼�������Һ��ϴ��ƿ���ռ� |
D | MnO2��KCl | ���� | �ܽ⡢���ˡ�ϴ�ӡ���ɡ������ᾧ |
A. A B. B C. C D. D