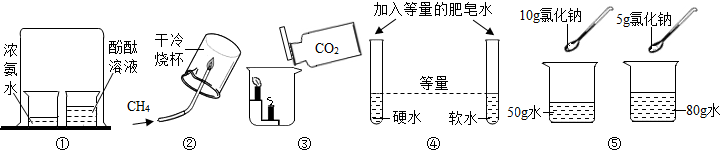
��Ŀ����
10�����ÿ����е��������ƹ����Ƿ���ʣ�����������̽������1�����ÿ����е��������ƹ��壺������ˮ�ݶ����⣻������CO2�����ʣ�
��2��Ϊ����֤���������Ƿ���ʣ��ֱ�ѡ�����������Լ�����ʵ�飺
һ��ϡ�����������������Һ�������Ȼ�����Һ
������һ��Ҫ������������Ʒ�����Һ�ٽ���ʵ����Լ��Ƕ�������������������Һ���Ȼ�����Һ�������������Ʒ�Ѿ����ʣ�
���Լ���ʵ���з�����Ӧ�Ļ�ѧ����ʽΪNa2CO3+CaCl2�TCaCO3��+2NaCl��
��ʵ����������ʵ���һ���ྻ���ձ��У��õ���ɫ������Һ������Һ��һ�����е�������NaCl��CaCl2��
���� ���⿼���������Ƶ����ʣ��������������׳��⣬��������еĶ�����̼������ѧ��Ӧ������̼���ƣ���˼����Ƿ���ʼ������Ƿ����̼������ӣ��ݴ˽��з������
��� �⣺
��1�������������������ˮ�����⣬�������̼��Ӧ����̼���ƺ�ˮ������ʱ���������л���̼���ƣ�
��2���ټ�������������Һ���Ƿ���̼���ƣ�������Һ�м�������ϡ�����ϡ���ᣬ������ð�����ɼ��麬��̼���ƣ�������Һ�еμ�ijЩ����Һ���������ƻ�����������Һ���г����������ɼ��麬��̼���ƣ�������Һ�м���ijЩ����Һ�����Ȼ��ƻ�����ƣ��г����������ɼ��麬��̼���ƣ�
����һ��Ҫ������������Ʒ�����Һ�ٽ���ʵ����Լ��Ƕ�������������������Һ���Ȼ�����Һ����
������������Һ�ͺ�̼���Ʒ�Ӧ����̼��Ƴ������������ƣ��Ȼ�����Һ��̼���Ʒ�Ӧ����̼��Ƴ������Ȼ��ƣ���Ӧ�Ļ�ѧ����ʽΪNa2CO3+CaCl2�TCaCO3��+2NaCl��
��ʵ����������ʵ���һ���ྻ���ձ��У��õ���ɫ������Һ������Һ��һ�����е�������NaCl��CaCl2
�ʴ�Ϊ��
��1��CO2
��2��
�ٶ�������������������Һ���Ȼ�����Һ����
��Na2CO3+CaCl2�TCaCO3��+2NaCl
��NaCl��CaCl2��
���� Ӧ��Ϥ���ʵ����ʼ���صĻ�ѧ��Ӧ����ʽ����ע���ʵ����ԣ���Ϥ���鳣�����ӵķ�����

| A�� | ���̼ӹ��������ȥCO | B�� | ���ͼ�����ֹƶѪ | ||
| C�� | ʳ�μӵ��ֹ��״���״� | D�� | ���ˮ����ˮ��Ӳ�� |
| A�� | NaOH��FeCl3��MgSO4��BaCl2��KCl | B�� | AgNO3��HCl��Na2CO3��KCl��CaCl2 | ||
| C�� | Ba��NO3��2��NaCl��Na2SO4��H2SO4��HCl | D�� | NaCl��BaCl2��CuSO4��NaOH��KNO3 |
���������ϡ�
��Na2S4����ʱ����������е�ˮ������Ӧ�����������ж��������⣨ˮ��Һ�����ԣ���
����������ƣ�Na2S2O3��������ˮ����������NaOH��Һ����Ӧ��
��ʵ����̡�
��ȡ0.5g̼���ơ�0.2g��ۻ�Ϻ������Թ��У����루��ͼ1��ʾ���г�װ������ȥ�����Ƶ�Na2S4����ӦΪ��4Na2CO3+12S+X$\frac{\underline{\;\;��\;\;}}{\;}$2Na2S4+2Na2S2O3+4CO2��X�Ļ�ѧʽΪO2��պ��ŨNaOH��Һ�������������������ɵĶ�����̼������ж����������壮
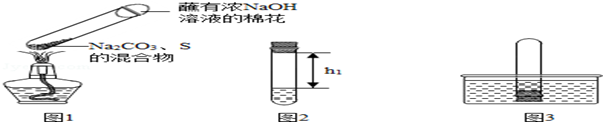
����ȴ��ȡ����������һ��ʱ�䣬������Թ��м���10mLˮ��Ѹ���������������������Һ�����������صľ��룬��¼����h1����ͼ2��ʾ����
�۽����Թܲ���ˮ�У���ͼ3��ʾ���������������۲쵽ˮ�����Թ��ڣ�����ˮ�����ԼΪ�Թ��ڿ�����������֮һ���л�ɫ�������ɣ��������������Թ�ȡ������ת����������Һ�����������صľ��룬��¼����h2��������h2��h1=4��5��
�ܰ��բ١������ظ�ʵ��2�Σ�3��ʵ���������±���ʾ��
| ��1�� | ��2�� | ��3�� | |
| h1/cm | 11.0 | 11.4 | 11.6 |
| h2/cm | 8.7 | 9.1 | 9.2 |
���ݵ�1��ʵ�����ݣ�����������������������Ϊ20.9%�������ȷ��0.1%����
����˼�����ۡ�
��ʵ����̢��У�ȡ������δ����һ��ʱ�伴���к�����������Ӱ��ⶨ�������˵���к�Ӱ�죬���������ɣ�ƫС����Ϊ�ڣ�1���еķ�Ӧ���Թ��е����������ľ���������̼��Ȼ�����գ�����ȡ�������Թ��ж�����̼����û�ж��ٸı䣨ԭ��û�У�����ֻ��0.04%�������Բ����ǰ�����������ֱ�ӿ��Ժ��������ԭ��û�У�����20%������������һ��ʱ�����Թ������������ȿ�����ƫ�٣�����h2ƫ���������ս��Ӧ��ƫС�ģ���