��Ŀ����
12��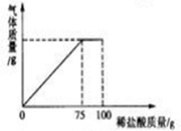 ij����ѧϰС���ͬѧ�ǣ�Ԥ�ⶨһ������ij��ʯ��ʯ��Ʒ��̼��Ƶ�����������������ʵ�飺
ij����ѧϰС���ͬѧ�ǣ�Ԥ�ⶨһ������ij��ʯ��ʯ��Ʒ��̼��Ƶ�����������������ʵ�飺��1������������������Ϊ7.3%��ϡ���ᣮ
������100g������������Ϊ7.3%��ϡ���ᣬ��36.5%����������20g��
����ȡ������ˮʱ������Ҫ����Ͳ��Һ�尼Һ�����ʹ�����ˮƽ���ٶ���Һ��������������Һ�嶼�����ձ��У����������ƣ����ò��������裬ʹ��Һ��Ͼ��ȣ�
��2������1�������ƺõ�ϡ�������ʢ��ʯ��ʯ��Ʒ���ձ��У�������������������ϡ�����������ϵ��ͼ��ʾ������ʯ��ʯ��Ʒ�е����ʲ���ϡ���ᷴӦ�������㣺��ʯ��ʯ��Ʒ��̼��Ƶ�������
���� ��1������ϡ��ǰ�����ʵ���������������𣬸�����Ͳ��ʹ�÷�������Һ�����Ʒ�����������
��2�����ݻ�ѧ����ʽ�Ļ����������صļ�����з������ɣ�
��� �⣺��1��������ƿŨ����������100g������������Ϊ7.3%��ϡ���ᣬ����ȡ����Ũ�����������$\frac{100g��7.3%}{36.5%}$=20g��
����ȡ������ˮʱ������Ҫ����Ͳ��Һ�尼Һ�����ʹ�����ˮƽ���ٶ���Һ��������������Һ�嶼�����ձ��У��ò��������裬ʹ��Һ��Ͼ��ȣ�
��2���� �μӷ�Ӧ��̼��Ƶ�����Ϊx
CaCO3+2HCl=CaCl2+H2O+CO2��
100 73
x 75g��7.3%
$\frac{100}{x}$=$\frac{73}{75g��7.3%}$
x=7.5g
�ʴ�Ϊ����1����20��
����ʹ����ձ���
��2����ʯ��ʯ��̼��Ƶ�����Ϊ7.5g��
���� ������Ҫ���������ʵ�������������ؼ�������ݻ�ѧ����ʽ�ļ��㣬�ѶȲ������ʱҪϸ�ļ��㣮

��ϰ��ϵ�д�
 �������Ӧ���⼯ѵϵ�д�
�������Ӧ���⼯ѵϵ�д� �ۺ��Բ�ϵ�д�
�ۺ��Բ�ϵ�д�
�����Ŀ
3�����л����У����ڸ��Ϸ��ϵ��ǣ�������
| A�� | NH4NO3 | B�� | NH4H2PO4 | C�� | K2SO4 | D�� | Ca��H2PO4��2 |
20�����й���ȼ�պ�������������ȷ���ǣ�������
| A�� | ���촵����������Ϊʹ������������� | |
| B�� | ���ڷ�������ʱ����óȻ���Ŵ� | |
| C�� | ��ȼ���������Ӵ����Խ��ȼ��Խ���� | |
| D�� | �����͡����������Ż��ʹ�øɷ������ |
1���÷��ӵĹ۵����������Ľ��ʹ�����ǣ�������
| ѡ�� | ���� | ���� |
| A | ˮ��ɱ��������� | ���Ӽ����� |
| B | ����IJ�������ԭ | ���Ӽ���ڳ��� |
| C | �ŵ�·�����˵Ļ��� | ���Ӳ�ͣ���˶� |
| D | �������ƽ����Ǧ���ѹ��������� | ���Ӽ�������� |
| A�� | A | B�� | B | C�� | C | D�� | D |
11����ͼ��ʾ��ʵ������ܴﵽʵ��Ŀ���ǣ�������
| A�� |  ��ȼ�ŵ�ľ�����������̼ | B�� |  ��������ʳ��ˮ | ||
| C�� |  �������̼���Ʒ�Ӧ��֤�����غ� | D�� |  ����Ͳ����ȡ46mL��ˮ |