��Ŀ����
��֪���ٳ��³�ѹ�£�������O3��Ϊ��ɫ���壻��O3�к�ǿ������ ��Ũ�ȳ���ʱ�������ж������ã����ڵ��������O2��ת��ΪO3��N2Ҳ����O2��Ӧ����KI�ᱻǿ����������Ϊ���ʵ⣬���ʵ������۱�������Ʒ����ҺΪ��ɫ����ǿ���������Ϊ��ɫ��
��Ũ�ȳ���ʱ�������ж������ã����ڵ��������O2��ת��ΪO3��N2Ҳ����O2��Ӧ����KI�ᱻǿ����������Ϊ���ʵ⣬���ʵ������۱�������Ʒ����ҺΪ��ɫ����ǿ���������Ϊ��ɫ��
ʵ�����ṩ����װ�ú�ҩƷ�������O3���Ʊ���������֤ʵ�顣

��1������װ�õ�����˳��Ϊ_______��_______��_______��______
��2��B�з�����Ӧ�Ļ�ѧ����ʽΪ________________________________
��3��ʵ�鿪ʼʱ��Ҫ��ͨһ��ʱ���������ٺ��ϵ�Դ���ؿ�ʼ����O3��ԭ���ǣ�
______________________________________________________________________
��4�����Ͽ���һ��ʱ���Dװ���г��ֵ�������_______________________________
��5���Ӱ�ȫ�������Ƕȿ���Ӧ���ӵ�װ����__________________________װ��
��1���ڢܢ٢�
��2��2H2O2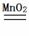 2H2O+O2��
2H2O+O2��
��3���ž���������ֹN2�� O2��ӦӰ��O3�����ɺ�����֤��
O2��ӦӰ��O3�����ɺ�����֤��
��4���������ݣ���Һ������ƿ�ϲ�����ɫ���塣
��5��β�����գ�������
����2�֣�

 ��ʦָ����ĩ��̾�ϵ�д�
��ʦָ����ĩ��̾�ϵ�д�
|
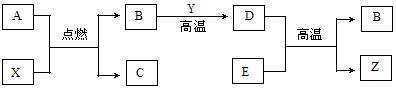
 ������������Ʒ�����ݲ��������ȷ���л������ɣ���֪��C�е�����ͭ��ȷ���л����е�̼Ԫ����ȫת��Ϊ������̼��A�еķ�ӦΪ��2H2O=====2H2O+O2����
������������Ʒ�����ݲ��������ȷ���л������ɣ���֪��C�е�����ͭ��ȷ���л����е�̼Ԫ����ȫת��Ϊ������̼��A�еķ�ӦΪ��2H2O=====2H2O+O2���� 
�ش��������⣺
��1��Aװ����a����������________��b��ʢ�ŵ�������___________��
��2����ȥ��Bװ�û��ʲôԪ�صIJ��������Ӱ�죿______����ʹ�������_______���ƫ��ƫС������
��3��Eװ������ʢ���������ѡ��__________����ѡ�������������Һ��������������Һ����ˮ��
��4����ȷ��ȡ1.12g��Ʒ����Ʒֻ��C��H��O����Ԫ���е����ֻ����֣��������ȼ�գ�Dװ������1.44g��Eװ������3.52g������л�����Ʒ��������Ԫ��Ϊ_______________��
 ___������
___������