��Ŀ����
23���ᡢ�����ճ�������������й㷺��Ӧ�ã�д�����б仯�Ļ�ѧ��Ӧ����ʽ
��1����ʯ�ұ�¶�ڿ�����ʱ�䳤�˻���ʣ���ʯ�ҷ�ˢ���ݣ�����ס������⣮
��Ӧ�Ļ�ѧ����ʽ��
��2��θ��ƽ�к�����������������������θ����࣮
��Ӧ�Ļ�ѧ����ʽ��
��3������м�����������������ͭ��Һ�ķ�Һ��
��Ӧ�Ļ�ѧ����ʽ��
��1����ʯ�ұ�¶�ڿ�����ʱ�䳤�˻���ʣ���ʯ�ҷ�ˢ���ݣ�����ס������⣮
��Ӧ�Ļ�ѧ����ʽ��
Ca��OH��2+CO2=CaCO3��+H2O
����2��θ��ƽ�к�����������������������θ����࣮
��Ӧ�Ļ�ѧ����ʽ��
Al��OH��3+3HCl=AlCl3 +3H2O
����3������м�����������������ͭ��Һ�ķ�Һ��
��Ӧ�Ļ�ѧ����ʽ��
Fe+H2SO4 =FeSO4 +H2����CuSO4 +Fe=FeSO4 +Cu
�����������ݷ�Ӧ�������Լ���Ӧ��������ȷ��д��ѧ��Ӧ����ʽ��ע��һ��Ҫ��ƽ��
����⣺��1����ʯ�ұ�¶�ڿ�����ʱ�䳤�˻���ʣ���ʯ�ҷ�ˢ���ݸ���ס������⣬������Ϊ��ʯ�ҺͶ�����̼��Ӧ������̼��ƺ�ˮ����Ӧ�Ļ�ѧ����ʽΪ��Ca��OH��2+CO2=CaCO3��+H2O��
��2��θ��ijɷ����ᣬ��θ��ƽ�к��е�������������θ����࣬�����������������Ӧ�����Ȼ�����ˮ����Ӧ�Ļ�ѧ����ʽΪ��Al��OH��3+3HCl=AlCl3 +3H2O��
��3�����������ͭ��ǰ�棬���������ܺ����ᷴӦ�ų�������Ҳ�ܺ�����ͭ��Һ��Ӧ��������������ͭ����Ӧ�Ļ�ѧ����ʽΪ��Fe+H2SO4 =FeSO4 +H2����CuSO4 +Fe=FeSO4 +Cu��
�ʴ�Ϊ����1��Ca��OH��2+CO2=CaCO3��+H2O����2��Al��OH��3+3HCl=AlCl3 +3H2O����3��Fe+H2SO4 =FeSO4 +H2����CuSO4 +Fe=FeSO4 +Cu��
��2��θ��ijɷ����ᣬ��θ��ƽ�к��е�������������θ����࣬�����������������Ӧ�����Ȼ�����ˮ����Ӧ�Ļ�ѧ����ʽΪ��Al��OH��3+3HCl=AlCl3 +3H2O��
��3�����������ͭ��ǰ�棬���������ܺ����ᷴӦ�ų�������Ҳ�ܺ�����ͭ��Һ��Ӧ��������������ͭ����Ӧ�Ļ�ѧ����ʽΪ��Fe+H2SO4 =FeSO4 +H2����CuSO4 +Fe=FeSO4 +Cu��
�ʴ�Ϊ����1��Ca��OH��2+CO2=CaCO3��+H2O����2��Al��OH��3+3HCl=AlCl3 +3H2O����3��Fe+H2SO4 =FeSO4 +H2����CuSO4 +Fe=FeSO4 +Cu��
������������Ҫ���黯ѧ����ʽ����д�ȷ����֪ʶ����д��ѧ����ʽʱҪע����ѭ�����غ㶨�ɣ�

��ϰ��ϵ�д�
�����Ŀ
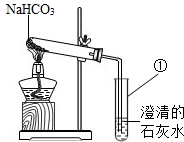 ��2012?�ݰ����ʼ죩��̽��ʵ��1��̼��������Һ������ԣ�
��2012?�ݰ����ʼ죩��̽��ʵ��1��̼��������Һ������ԣ�