��Ŀ����
���������������(���к�������)��84����Һ(���к����㰱����)���ʹ�ã����ᷢ����Ӧ��������������Ӧ�Ļ�ѧ����ʽΪ2HCl+NaClO=NaCl+Cl2+H2O��
(1)������Ӧ�У�����Ԫ�ػ��ϼ���ߵ�������_____ (����ĸ)��
A.HCl B.NaClO C.NaCl D.Cl2
(2)����ˮ��������������Ȼˮ����ɱ���������ڴ�֮ǰ��Ҫ����������______�Ⱦ��������������н���ˮ��Ӳ�ȵij��÷�����______��
��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ
�±���һЩʳ���pH��θ�����IJ��˿ո�ʱ����ʳ�õ��ǣ� ��
���� | ���� | ���� | ţ�� | ������ |
pH | 3��4 | 3.5��4.5 | 6.3��6.6 | 6.8��8.0 |
A������ B������ C��ţ�� D��������


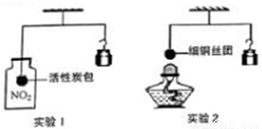
 Fe3O2 B. CO+CuO
Fe3O2 B. CO+CuO  Cu+CO2
Cu+CO2 ��ȡҺ����� B.
��ȡҺ����� B. 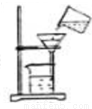 ���� C.
���� C. 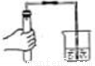 ���װ�õ������� D.
���װ�õ������� D. 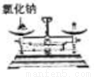 �����Ȼ���
�����Ȼ���