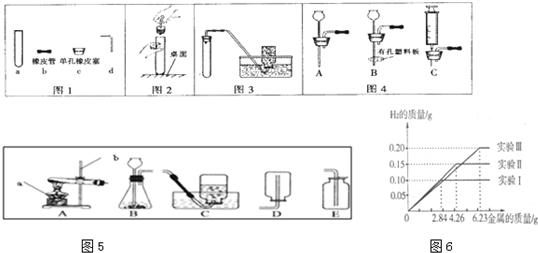
��Ŀ����
��ѧ��һ����ʵ��Ϊ�����Ŀ�ѧ��
������ͼ��ʾʵ�飬�ش��������⡣
������ͼ��ʾʵ�飬�ش��������⡣
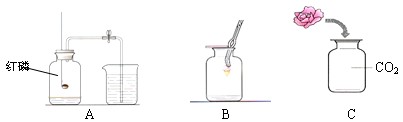
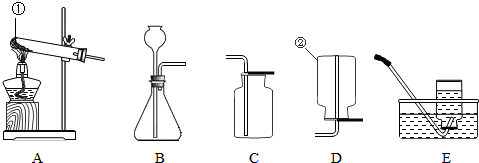
��1��AΪ�ⶨ����������������ʵ��װ�ã��йط�Ӧ�Ļ�ѧ����ʽΪ_________________________��ͨ����ʵ��ɵó�����Լռ�����������________�Ľ��ۡ�
��2������ƿB�е�������˿�������о���ȼ�գ�______________���ų��������ȣ��к�ɫ�������ɡ��÷�Ӧ�Ļ�ѧ����ʽΪ____________________________��
��3��ȡһ����ʯ����ҺȾ����ɫ�ĸ���С������С����һ����ˮ��ʪ�����뼯��ƿC�У��۲쵽��������____________________________��
��2������ƿB�е�������˿�������о���ȼ�գ�______________���ų��������ȣ��к�ɫ�������ɡ��÷�Ӧ�Ļ�ѧ����ʽΪ____________________________��
��3��ȡһ����ʯ����ҺȾ����ɫ�ĸ���С������С����һ����ˮ��ʪ�����뼯��ƿC�У��۲쵽��������____________________________��
��1��4P+5O2 2P2O5��1/5(��21%)
2P2O5��1/5(��21%)
��2���������䣻3Fe+2O2 Fe3O4
Fe3O4
��3����ʪ�IJ������ϱ��
 2P2O5��1/5(��21%)
2P2O5��1/5(��21%)��2���������䣻3Fe+2O2
 Fe3O4
Fe3O4��3����ʪ�IJ������ϱ��

��ϰ��ϵ�д�
�����Ŀ
 20����ѧ��һ����ʵ��Ϊ�����Ŀ�ѧ���淶��ʵ�������ʵ��ɹ���ǰ�ᣬ��ش�
20����ѧ��һ����ʵ��Ϊ�����Ŀ�ѧ���淶��ʵ�������ʵ��ɹ���ǰ�ᣬ��ش�