��Ŀ����
�⻯�ƹ����ǵ�ɽ�˶�Ա���õ���Դ�ṩ����ij̽��С���ͬѧͨ���������ϵ�֪���⻯�ƣ�CaH2����ˮ��Ӧ�����������ƺ�������̽��С���ͬѧ��һ������CaH2����Na2CO3��Һ�У���ַ�Ӧ����ˣ��õ���������Һ�������������ijɷ���̼��ƣ�
��������⡿��Һ�����ʵijɷ���ʲô��
����������衿
����һ��NaOH �������NaOH��Ca��OH��2
��������NaOH��Na2CO3 �����ģ�NaOH��Na2CO3��Ca��OH��2
�������ۣ����һ����Ϊ�����IJ����������û�ѧ����ʽ˵��ԭ��__________________��ʵ����֤��
 ����˼����չ��������NH4Cl����Һ�м�һ����CaH2����ַ�Ӧ�������������ǡ�_______���ڵ�ɽ�˶�Ա����CaH2��Ϊ��Դ�ṩ������������ȣ����ŵ���__________________��
����˼����չ��������NH4Cl����Һ�м�һ����CaH2����ַ�Ӧ�������������ǡ�_______���ڵ�ɽ�˶�Ա����CaH2��Ϊ��Դ�ṩ������������ȣ����ŵ���__________________��
���𰸡���1��Na2CO3+Ca��OH��2=CaCO3��+2NaOH����2���������ޱ仯��������ݣ�
��3������������������Я����ȫ
����������1������������衿�������е�̼�������������Ʋ��ܹ��棬���Ӧ����̼��ư�ɫ�������������ƣ��ʲ�������
��2����ʵ����֤��ʵ��һ������������˵��û���������ƣ��������������ʵ������۲쵽���ޱ仯��������ݵ�����ʱ��˵����Һ�к����������ƺ�̼���ƣ���������������
��3������˼����չ����NH4Cl����Һ�м�һ����CaH2��������������Ͱ�������Ϊ�⻯�ƣ�CaH2����ˮ��Ӧ�����������ƺ��������Ȼ�狀��������Ʒ�Ӧ�����Ȼ��ƺ�ˮ�Ͱ�����CaH2��Ϊ��Դ�ṩ�����ŵ���Я�����㰲ȫ���������������ȡ�������㡣

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д���ʮһ�����Ǻ��������õ����ͣ�����ʵʩ�����������ս���γ��µķ�չ��֣�����ѧ��Դ���ۺ����ý����ص㷢չ����֮һ��
��1����ˮɹ�Σ�����ˮ���������أ�����ɹ������һ���̶�ʱ������ᾧ�أ�������ɹ����ˮ�ͻ��Ϊʳ�εı�����Һ����ɹ�ͻ����������Σ�ʣ���Һ���ΪĸҺ��Ҳ��±ˮ����
1L��ˮ����Ũ�������������������ε��������������λ��g����ϵ���±���ʾ��
| ��ˮ�ܶȣ�g/mL�� | CaSO4 | NaCl | MgCl2 | MgSO4 |
| 1.20 | 0.91 | |||
| 1.21 | 0.05 | 3.26 | 0.004 | 0.008 |
| 1.22 | 0.015 | 9.65 | 0.01 | 0.04 |
| 1.26 | 0.01 | 2.64 | 0.02 | 0.02 |
| 1.31 | 1.40 | 0.54 | 0.03 |
I.��ˮɹ���� ��ѡ���������ѧ�����仯����������. �ȹ��̡�
II.���õ����ȽϸߵĴ��κͺ�Ũ�Ƚϸߵ�±ˮ��±ˮ���ܶ�Ӧ������ʲô��Χ ��ԭ���� ��
��2����ˮ��þ����ˮɹ�κ�����ĸҺ��±ˮ���ͱ��ǣ���Ҫ�ɷ���CaCO3��Ϊԭ����þ���乤����������ͼ��ʾ��
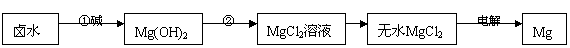

I.��������ӵļ�Ӧ���� ��ѡ��������ơ���ʯ���顱��������ڷ�Ӧ�Ļ�ѧ����ʽ�� ����Ҫ��֤þ.��.ͭ�Ļ��˳��ѡ����˿��Ҫѡ�� . ������Һ��
II.Mg(OH)2���ȷֽ�����MgO��H2O��MgO�Ǹ��۵�Ļ����ijЩ��ȼ�����谭ȼ�յ����ʣ�����Ч�ɷ���Mg(OH)2������ΪMg(OH)2�ܹ���ȼ��ԭ������� ��ѡ����ţ�
A.Mg(OH)2�ֽ���Ҫ���մ������ȣ������˿�ȼ����Ż��
B.���ɵ�����þ�����ڿ�ȼ����棬�����˿���
C.���ɵ�ˮ���������˿�ȼ����Χ����Ũ��
D.��Ӧ�����Ƚ����˻����¶ȣ�ʹ��ȼ�ﲻ�״ﵽ�Ż��
ͬѧ�����á����ᡢ����李��������ơ�̼��ء��������ʵ���Һ����̽��ѧϰ��������и��ʣ�
��ʵ��̽����
��1��A��B��C��D����ͬѧ�ֱ����������ʵ��

д�����е�������ѧ����ʽ��
��A�еķ�Ӧ
��D�еķ�Ӧ
���ھ�ʵ�ʡ�
��ͼ��A�鷴Ӧ���۹���ʾ��ͼ�û�ѧ��Ӧ��ʵ�ʿ��Ա�ʾΪ 
 ��ʵ�����á�
��ʵ�����á�
������������ͬѧ����Һ����ͬһ���ձ��У��ٽ���Һ���ˣ��õ���ɫ��Һ�Ͱ�ɫ����������ɫ��Һ�μ���ɫʯ���Լ�����Ϊ��ɫ��
��2������Һ�еİ�ɫ����һ���� ��������
��3����Һ��һ�������е�������
��4��Ϊ����֤�� ��Һ�п��ܺ��е������Ƿ���ڣ����������ʵ�������֤��
��Һ�п��ܺ��е������Ƿ���ڣ����������ʵ�������֤��
| ��Һ�п��ܺ��е����� | ʵ�鲽�� | ʵ������ | ʵ����� |
| �ٿ��ܺ��� | ��ȡ��Һ�μ� | �������� | ���и����� |
ʵ�����е�ҩƷһ��Ҫ�ܷⱣ�棬������ܻ�������Ӵ������ʡ�ij�о���ѧϰС���ͬѧ��ʵ����������ڿ����б��ʵĹ������ƣ�Na2O2)����ijɷֽ���̽����
[��������]��1��Na2O2�Ļ�ѧ���ʺܻ��ã�����ˮ��������̼��Ӧ����ط�Ӧ�Ļ�ѧ����ʽΪ��2Na2O2+2H2O=4NaOH+O2�� 2Na2O2+2CO2=2Na2CO3+O2��
��2��CaCl2��ҺΪ����
[����]I������ΪNa2O2��NaOH��Na2CO3�Ļ����
II������ΪNaOH��Na2CO3�Ļ����
III������ΪNaOH
IV������ΪNa2CO3
[ʵ��̽��]
| ʵ����� | ʵ������ | ʵ����� |
| ��ȡ�����������Թ��У���ˮ��ֱ����ȫ�ܽ� |
| ����I������ |
| ��ȡ�����ٵ���Һ���Թ��У��μ�CaCl2��Һ������ |
| ֤����Na2CO3���� |
| ��
|
| ֤����NaOH���� |
| �ۺ�����ʵ������˵������II�dz����ġ� |
[��˼����]��1��ʵ��������з�Ӧ�Ļ�ѧ����ʽΪ
��2��ʵ��������С��μ�CaCl2��Һ����������Ŀ����


 ��4��д������ȼ�յĻ�ѧ����ʽ_________________________________________��
��4��д������ȼ�յĻ�ѧ����ʽ_________________________________________��


 ����CO ��С��ͬѧ���л�������ʱ���֣������������糧����úΪȼ�ϣ���Χ����ľ�ѽ���ή�����糧�ŷŵķ�����Ҳ���ܺ�CO �������ռ��ķ�����Ʒ����ͼ��ʾװ��ǡ����Ϻ���м��飬��֪���е�ҩƷ����������ش��������⣺
����CO ��С��ͬѧ���л�������ʱ���֣������������糧����úΪȼ�ϣ���Χ����ľ�ѽ���ή�����糧�ŷŵķ�����Ҳ���ܺ�CO �������ռ��ķ�����Ʒ����ͼ��ʾװ��ǡ����Ϻ���м��飬��֪���е�ҩƷ����������ش��������⣺
 �����������Ӧ�Ļ�ѧ��Ӧ����ʽ����Ϊ ��
�����������Ӧ�Ļ�ѧ��Ӧ����ʽ����Ϊ ��
 �ƣ�
�ƣ�  ��
��