��Ŀ����
15��ʵ����������ʯ�ҡ�����ء��������̡�ϡ����ͳ���ʯ��ˮ����ص�������������Ʒ����ش��������⣺
��1����������ҩƷ������һ�������ƾ��Ƽ����Ʊ�ij�����壬��Ӧ�Ļ�ѧ����ʽ2KClO3$\frac{\underline{MnO_2}}{��}$2KCl+3O2����
��2������ҩƷ��ʯ�һ���̼��ƾͿ��Ʊ���������һ�����壬���������ķ����ǽ�����ͨ�뵽����ʯ��ˮ�У�������ʯ��ˮ��������Ƕ�����̼��
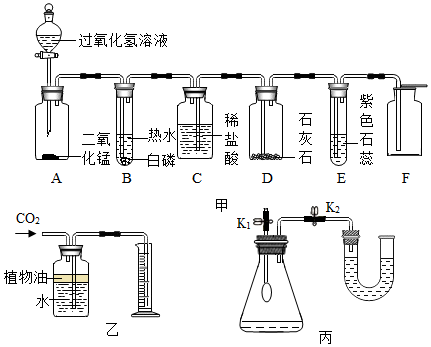
��3����û����ɵ���ȡ������̼��װ��ͼ����������
���� ��1��������ҩƷ��֪�����ü�������غͶ������̵Ļ����ķ�����ȡ��������Ҫ���ȣ����Ի���Ҫ�ƾ��ƣ��ݷ�Ӧԭ����д����ʽ��
��2��ʵ������ȡ������̼�ô���ʯ��ʯ��ʯ��ϡ���ᣬ���������̼�ó����ʯ��ˮ��
��3��ͼ2���嵼������Ҫ��˫�����ĵ��ܣ���Ҫ����©�������¶�Ҫ����Һ�����£���ֹ���ɵĶ�����̼�ӳ���©���ݳ���
��� �⣺��1��������ҩƷ��֪�����ü�������غͶ������̵Ļ����ķ�����ȡ������ҩƷ�����Թ��У�����Ҫ���ȣ����貹��ƾ��ƣ�������ڶ������̵Ĵ������¼��������Ȼ��غ���������Ӧ�Ļ�ѧ����ʽΪ��2KClO3$\frac{\underline{MnO_2}}{��}$2KCl+3O2����
��2��ʵ������ȡ������̼�ô���ʯ��ʯ��ʯ��ϡ�����ϣ����Կɽ���ʯ�һ�Ϊ����ʯ��ʯ��ʯ�����������Ƿ��Ƕ�����̼���ɽ���������ʯ��ˮ���۲��Ƿ����ǣ�
��3��ͼ2��˫����������Ҫ����©�������¶�Ҫ����Һ�����£���ֹ���ɵĶ�����̼�ӳ���©���ݳ�������ͼ����
�ʴ�Ϊ��
��1���ƾ��ƣ�2KClO3$\frac{\underline{MnO_2}}{��}$2KCl+3O2����
��2����ʯ�ң�����ʯ����ʯ��ʯ����������ͨ�뵽����ʯ��ˮ�У�������ʯ��ˮ��������Ƕ�����̼
��3��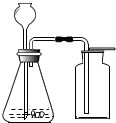 ��
��
���� ��ȷʵ������ȡ������ҩƷ��������ԭ����װ�õȣ��ǽ�����Ĺؼ������ܽ�����������

| A�� | ��Ȫˮ | B�� | �ӵ�ʳ�� | C�� | ��ˮ | D�� | ����ˮ |
| ѡ�� | ���� | ���ʣ������� | �����Լ������� | ���Ӳ������� |
| A | KCl��Һ | CaCl2 | ������Na2CO3��Һ | ���ˡ���ȥ���� |
| B | NaCl | NaOH | ���������� | ���������ᾧ |
| C | Cu��NO3��2��Һ | AgNO3��Һ | ����ͭ�� | ���˽�������� |
| D | ̼�� | ����ͭ�� | �ڿ����г������ |
| A�� | A | B�� | B | C�� | C | D�� | D |
| A�� | �������ᷴӦ����AlCl3��H2��������������ᷴӦ����FeCl3��H2 | |
| B�� | ̼���������ᷴӦ�������壬�������ᷴӦ�������������һ����̼���� | |
| C�� | ����Һ��ʹ��ɫ��Һ��죬������ʹ��ɫʯ�����һ���������Һ | |
| D�� | ����кͷ�Ӧ��ˮ���ɣ���ˮ���ɵķ�Ӧ��һ��������кͷ�Ӧ |
| A�� | Һ�� | B�� | �ɱ� | C�� | ���� | D�� | ���� |
| A�� | �� �Ȼ��� ʳ�� NaCl | |
| B�� | �� ̼������ С�մ� NaHCO3 | |
| C�� | ������ ������ ��ʯ�� CaO | |
| D�� | �� �������� �� �� NaOH |