��Ŀ����
20�����Ͳ�������Fe�۾��й㷺����;��������ͨFe�۸�����������Ӧ�����Ʊ�����������ͼ��ʾ����ش��������⣺
��1������Fe�������������Է�ȼ�����ɺ�ɫ���壬�˺�ɫ����Ļ�ѧʽΪFe3O4��
��2���벹����������Ʊ����յĻ�ѧ����ʽ��H2+FeCl2$\frac{\underline{\;����\;}}{\;}$Fe+2HCl�����Ʊ�����Fe�۵Ĺ��������У�N2�������DZ�������
��3���о���Ա��������Ƶõ�����Fe����Ʒ�л��������� FeCl2 ���ʣ�
��Ϊ��ȥ��Ʒ�е����ʣ������������£��ȼ�ˮ�ܽ⣬Ȼ���ٹ��ˡ�ϴ�ӡ���ɣ�
��ȡ��Ʒ10g���������������ᣬ�������� 0.3g����������Ʒ�е��� Fe ����������������д��������̣� ��Ӧ�Ļ�ѧ����ʽ��Fe+2HCl�TFeCl2+H2��
���� ��1����������������Ӧ�����������������ǣ���2�����ݷ�Ӧǰ�����ԭ���������Ŀ����͵�����ѧ�����ȶ����ǣ���3���ٸ�����������ˮ���Ȼ���������ˮ���л����ķ��룻�ڸ�����������������������������ٳ���10g�������������������
��� �⣺��1������������ȼ���������������������Ժ�ɫ������������������
��2����Ӧ��ķ����к���2����ԭ�ӡ�һ����ԭ�ӡ�2����ԭ�ӣ����������Ѿ�����һ����ԭ�ӣ���ȱ��2����ԭ�Ӻ�2����ԭ�ӣ����Բ����������2HCl��������ѧ�����ȶ����ڷ�Ӧ���𱣻��������ã�
��3������������ˮ���Ȼ���������ˮ�����Լ�ˮ�ܽ⣬�ٽ��й��ˡ�ϴ�Ӹ��T�ɣ�
��������0.7g������Ҫ�μӷ�Ӧ����������ΪX����
Fe+2HCl�TFeCl2+H2��
56 2
X 0.3g
���ݣ�$\frac{56}{x}=\frac{2}{0.3g}$
���X=8.4g��
$\frac{8.4g}{10g}$��100%=84%��
�ʴ�Ϊ����1��Fe3O4��
��2��2HCl����������
��3���ٹ��ˣ�
��������0.7g������Ҫ�μӷ�Ӧ����������ΪX����
Fe+2HCl�TFeCl2+H2��
56 2
X 0.3g
���ݣ�$\frac{56}{x}=\frac{2}{0.3g}$
���X=8.4g��
$\frac{8.4g}{10g}$��100%=84%��
���� �����ؼ���֪����ȼ�յIJ������������������ܹ�������������غ㶨�ɽ��ʵ�����⣬֪�������Թ����Һ�����ķ�������Ϥ���ݷ���ʽ�ļ��㷽����

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�| A�� |  ú����ұ�� | B�� |  ʯī���糵��ˢ | ||
| C�� |  ��ʯ�������к��������� | D�� |  ����������ֲ����� |
| A�� | NaOH��Һ��CuSO4��Һ | B�� | NaOH��Һ��ϡ���� | ||
| C�� | BaCl2��Һ��CuSO4��Һ | D�� | CuSO4��Һ������ |
| 3Li � | 4Be �� | 5B �� | 6C ̼ | 7N �� | 8O �� | 9F �� | 10Ne �� |
| 11Na �� | 12Mg þ | 13Al �� | 14Si �� | 15P �� | 16S �� | 17Cl �� | 18Ar � |
��2����Ԫ������Ԫ�ػ�ѧ�������ƣ���Ҫ����Ϊԭ�ӽṹ����������������6��
��3����1��8������Ԫ����ɣ���8��Ԫ�س�-1�۵����ʵĻ�ѧʽΪH2O2��
| A�� | �ڻ�������� | B�� | �ڻ����˶� | C�� | ������ͨ���� | D�� | �ؼҺ���ϴ�ڱ� |
| A�� | �����ڿ�����ȼ�ղ��������������������� | |
| B�� | ϸ��˿�ڿ����о���ȼ�գ��������䣬���ɺ�ɫ���� | |
| C�� | ���ڿ�����ȼ�գ���������������ɫ���� | |
| D�� | ������̼ͨ������ʯ��ˮ�У�ʯ��ˮ����� |
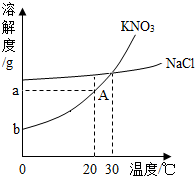 ��ͼΪ����غ��Ȼ��Ƶ��ܽ�����ߣ��ش��������⣺
��ͼΪ����غ��Ȼ��Ƶ��ܽ�����ߣ��ش��������⣺