��Ŀ����
14�������ȡ��������֤�������ʵ�ʵ�����һƿ��ɫҺ�壬���ǩֻ�ܿ���һ����ˮ���֣���Һ������ǣ�������ˮ����˫��ˮ������������Һ������ʯ��ʯ����1�����ƿҺ����ͨ��CO2ʼ��δ�۲쵽Һ�����ǣ���Һ��һ������ʯ��ˮ��
��2�������ƿҺ���м���MnO2���۲쵽���ҷ�Ӧ�������������ݣ���Һ����˫��ˮ��
��3����ʵ�飨1���ͣ�2���IJ����д���һ����ͬ��������ָ�����������û��ȡ����ֱ������Լ�ƿ�м����Լ�����
���� ��1�����ݶ�����̼��ʹ�����ʯ��ˮ����ǽ��
��2�����ݹ���������Һ�Ͷ������̻�ϻ������������
��3������ʵ�������ע��������з������
��� �⣺
��1��������̼��ʹ�����ʯ��ˮ����ǣ����ƿҺ����ͨ��CO2ʼ��δ�۲쵽Һ�����ǣ���Һ��һ������ʯ��ˮ��
��2�������ƿҺ���м���MnO2���۲쵽���ҷ�Ӧ�������������ݣ���Һ���ǹ���������Һ��˫��ˮ����
��3��ֱ������Լ�ƿ�м����Լ��������ҩƷ�˷ѣ�
�𰸣�
��1��ʯ��ˮ��
��2��˫��ˮ��
��3��û��ȡ����ֱ������Լ�ƿ�м����Լ�����
���� ���⿼�������ʵ����ʵļ��飬��ɴ��⣬�������ݱ�ǩ�ṩ����������ص㼰��������Ӽ���ķ������У�

��ϰ��ϵ�д�
�����Ŀ
4����ҵ���õ�ⱥ��ʳ��ˮ�ķ��������ռ�����������Ȼ�����Ʒ���䷴Ӧԭ��Ϊ��2NaCl+2H2O$\frac{\underline{\;ͨ��\;}}{\;}$2NaOH+H2��+Cl2����ij�о���С����Ƶõ��ռ���Ʒ����������̽����
��������⡿�ռ���Ʒ�п��ܺ�����Щ���ʣ�
�����롿
����һ�����ܺ���̼���ƣ������������CO2+2NaOH=Na2CO3+H2O���û�ѧ����ʽ��ʾ����
����������ܺ����Ȼ��ƣ�������������ڻ��������У�ԭ������������ȫת��Ϊ��Ʒ��
��ʵ��̽�������������̽��ʵ�飺
��������˼������Ϊ��ʵ�鲽����еμӹ���ϡ�����Ŀ���dz�ȥ��Ʒ�е�̼���ƣ�
��������⡿�ռ���Ʒ�п��ܺ�����Щ���ʣ�
�����롿
����һ�����ܺ���̼���ƣ������������CO2+2NaOH=Na2CO3+H2O���û�ѧ����ʽ��ʾ����
����������ܺ����Ȼ��ƣ�������������ڻ��������У�ԭ������������ȫת��Ϊ��Ʒ��
��ʵ��̽�������������̽��ʵ�飺
| ���� | ʵ�鲽�� | ʵ������ | ʵ����� |
| ����һ | ��ȡ�����ռ���Ʒ���Թ��У���ˮ�ܽ⣬�����еμӹ���ϡ���� | �����ݲ��� | ��Ʒ�к���̼���� |
| �ڽ�������в���������ͨ������ʯ��ˮ�� | ����ʯ��ˮ����� | ||
| ����� | ��������õ���Һ�еμӼ�����������Һ | ������ɫ���� | ��Ʒ�к����Ȼ��� |
5��úȼ�ղ����ķ����к���SO2����NaOH��Na2SO3��Һ�ɽ�90%���ϵ�SO2���ա��漰�ķ�Ӧ�У���2NaOH+SO2�TNa2SO3+H2O ��Na2SO2+SO2+H2O�T2NaHSO3 ��NaHSO3+NaOH�TNa2SO3+H2O ����˵���У�����ȷ���ǣ�������
| A�� | NaHSO3������ | |
| B�� | ��ѭ��ʹ�õ�������Na2SO3 | |
| C�� | ��Ӧ�ڷ������ǻ��Ϸ�Ӧ | |
| D�� | ��Ӧ��˵��SO2��CO2�����ƵĻ�ѧ���� |
6�������е�������Ʒ��ʹ�õ���Ҫ���Ϸ����л��ϳɲ��ϵ��ǣ�������
| A�� | ����ִ��� | B�� | �մɲ;� | C�� | ����Χȹ | D�� | ������ |
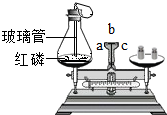 ��ѧ���ϣ�ͬѧ������ͼ��ʾʵ��̽�������غ㶨�ɣ�ͼ�з�Ӧ�Ļ�ѧ����ʽ��4P+5O2$\frac{\underline{\;��ȼ\;}}{\;}$2P2O5������ı仯������ȱ����С����С��ͬѧ����ʦ��Ҫ��淶���ʵ�飬����ʵ��ǰ����ƽ��ָ�붼ָ��b����������С��ͬѧ����ƽָ��ʵ��ǰָ��b����ʵ���ָ��c�������ܵ�ԭ���������û�еȵ���ȴ��ƿ��ȴ�ͳ�����дһ��������ɣ���С��ͬѧ�淶�˲����õ�������С��ͬ���Ľ����ʵ���ͬѧ�Ǵ��۽Ƕȷ����˻�ѧ��Ӧ���������غ㶨�ɵ�ԭ���ǻ�ѧ��Ӧǰ��ԭ�ӵ����ࡢ��Ŀ���������䣮
��ѧ���ϣ�ͬѧ������ͼ��ʾʵ��̽�������غ㶨�ɣ�ͼ�з�Ӧ�Ļ�ѧ����ʽ��4P+5O2$\frac{\underline{\;��ȼ\;}}{\;}$2P2O5������ı仯������ȱ����С����С��ͬѧ����ʦ��Ҫ��淶���ʵ�飬����ʵ��ǰ����ƽ��ָ�붼ָ��b����������С��ͬѧ����ƽָ��ʵ��ǰָ��b����ʵ���ָ��c�������ܵ�ԭ���������û�еȵ���ȴ��ƿ��ȴ�ͳ�����дһ��������ɣ���С��ͬѧ�淶�˲����õ�������С��ͬ���Ľ����ʵ���ͬѧ�Ǵ��۽Ƕȷ����˻�ѧ��Ӧ���������غ㶨�ɵ�ԭ���ǻ�ѧ��Ӧǰ��ԭ�ӵ����ࡢ��Ŀ���������䣮

 ˮ������֮Դ��
ˮ������֮Դ��