��Ŀ����
���ڴ���ս��ʱ�ڣ��ҹ��Ϳ�ʼ������ʹ��������
��1��д����CO�ͳ�������Ҫ�ɷ�Fe2O3��Ϊԭ�������Ļ�ѧ����ʽ
��ʢˮ��������ˮ��Ӵ���һȦ�������⡣��������������� �������¼����ܽ�����ּӹ��������Ľ���˿�����������˲���ֵ� ������ĸ����
A����ʴ�� B����չ�� C�������� D��������
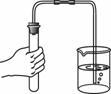
��2����ƴӷ�ˮ�л����������������ͭ��ʵ�鷽�����£� 
����X�� ��
д������ʵ�鷽�����йط�Ӧ�Ļ�ѧ����ʽ
�� ��  ��
��
��1��3CO+Fe2O3����2Fe+3CO2 ��ʪ����������ˮ�������Ӵ��� B
��2��Fe Fe+CuSO4=Cu+FeSO4 Fe+H2SO4=FeSO4+H2�� ����ѧ����ʽÿ��2�֣�����ÿ��1�֣�

 ��һ������ĩ�ٷֳ�̾�ϵ�д�
��һ������ĩ�ٷֳ�̾�ϵ�д�Ԫ�����ڱ��ǻ�ѧѧϰ���о�����Ҫ���ߣ���ͼ��Ԫ�����ڱ���һ���֡�
| ��A | 0 | ||||||||
| ��һ���� | 1 H �� 1.008 | 2 He �� 4.003 | |||||||
| ��A | ��A | ��A | ��A | ��A | ��A | ||||
| �ڶ����� | 3 Li � 6.941 | 4 Be �� 9.012 | 5 B �� 10.81 | 6 C ̼ 12.01 | 7 N �� 14.01 | 8 O �� 16.00 | 9 F �� 19.00 | 10 Ne �� 20.18 | |
| �������� | 11 Na �� 22.99 | 12 Mg þ 24.31 | 13 Al �� 26.98 | 14 Si �� 28.09 | 15 P �� 30.97 | 16 S �� 32.06 | 17 Cl �� 35.45 | 18 Ar� 39.95 |
 ��1��ij���Ľṹʾ��ͼΪ ��������Ԫ�����ڱ��е�__ _ _Ԫ�أ���
��1��ij���Ľṹʾ��ͼΪ ��������Ԫ�����ڱ��е�__ _ _Ԫ�أ���
�ĺ˵������_______��̼Ԫ�صĺ˵������_______���ɴ˿�֪��ͬԪ�صı���������_______
 ___________________��
___________________��
��2����ԭ�ӵĽṹʾ��ͼΪ  ���ڻ�ѧ��Ӧ����___________����á���ʧ����
���ڻ�ѧ��Ӧ����___________����á���ʧ����
���ӣ���_____________����������ǽ�������Ԫ�ء�
 ��Ӧ��������Һ��
��Ӧ��������Һ��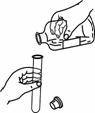
 ԭ�ӵ�ԭ�Ӻ˶������Ӻ����ӹ���
ԭ�ӵ�ԭ�Ӻ˶������Ӻ����ӹ���