��Ŀ����
��Դ��������������������ᷢչ������ء�
��1��Ŀǰ�������Ի�ʯȼ��Ϊ��Ҫ��Դ�������Ļ�ʯȼ�ϰ���ú��ʯ�ͺ� ��3�� ��
��2��Ϊ������Ⱦ �����ú�������ʣ��ɽ���ת��Ϊ��ȼ�����壬�˹��̿���Ϊ��̼��ˮ�ڸ����µķ�Ӧ������ʾ��ͼ������ʾ��
�����ú�������ʣ��ɽ���ת��Ϊ��ȼ�����壬�˹��̿���Ϊ��̼��ˮ�ڸ����µķ�Ӧ������ʾ��ͼ������ʾ��
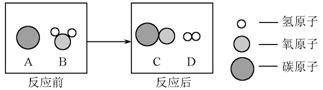
�÷�Ӧ�Ļ� ѧ����ʽΪ ��4�� ��������Ӧ����Ϊ ��5�� ��
ѧ����ʽΪ ��4�� ��������Ӧ����Ϊ ��5�� ��
������3��ȼ����ȫ�Ǽ�ͥ�����е�ͷ�ȴ��¡�Ϊ�˷�ֹȼ��й©������ȼ���м���������������ζ������ ��C2H5SH����һ��ȼ��й©�����ŵ���ζ�����۵ĽǶȷ�����ԭ���� ��6�� �������ȼ��ʱ���ɶ�����̼�����������ˮ������ȼ�յĻ�ѧ����ʽΪ ��7�� ��
��C2H5SH����һ��ȼ��й©�����ŵ���ζ�����۵ĽǶȷ�����ԭ���� ��6�� �������ȼ��ʱ���ɶ�����̼�����������ˮ������ȼ�յĻ�ѧ����ʽΪ ��7�� ��
��3����Ȼ�� ��4��C+H2O CO+H2 ��5���û���Ӧ ��6�������ڲ����˶�
CO+H2 ��5���û���Ӧ ��6�������ڲ����˶�
��7��2C2H5SH+9O2 4CO2+2SO2+6H2O
4CO2+2SO2+6H2O

��ϰ��ϵ�д�
�����Ŀ