��Ŀ����
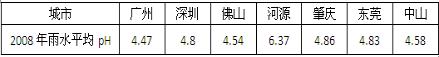
200һ����ʡ���ֵ���������ˮƽ��pH���±�����ش��������⣺
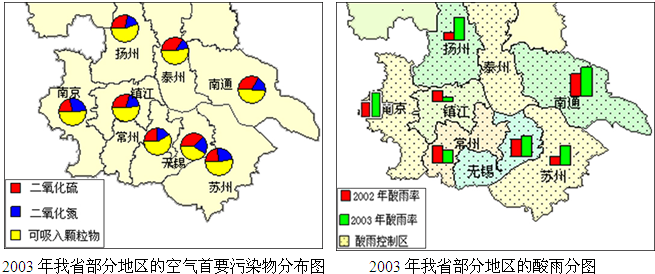
��1���ϱ��оٵ�5�������У�û��������Ⱦ�ĵ�����______�������������Ҫ������______������ţ���
��CO ��CO2��SO2��
��2�����������Ҫ�ɷ�ΪH2SO����HNO3����д�����л�ѧ����ʽ��
�ٺ�H2SO�������������ⷴӦ______��
�ں�HNO3��������������������ʯ�ҷ�Ӧ______��
| ���� | ���� | �Ͳ� | Ϋ�� | ̩�� | �ij� |
| 200һ����ˮƽ��pH | ��.��7 | ��.8 | ��.5�� | 6.37 | ��.86 |
��CO ��CO2��SO2��
��2�����������Ҫ�ɷ�ΪH2SO����HNO3����д�����л�ѧ����ʽ��
�ٺ�H2SO�������������ⷴӦ______��
�ں�HNO3��������������������ʯ�ҷ�Ӧ______��
��1��������ָPH��5.6����ˮ���ɱ��п�֪̩��û��������Ⱦ�������������Ҫ�����Ƕ��������̩�����ۣ�
��2������������������Ӧ������������ˮ�����Fe2O3+3H2SO4=Fe2��SO4��3+3H2O
��������������Ʒ�Ӧ��������ƺ�ˮ�����2HNO3+Ca��OH��2=Ca��NO3��2+2H2O
��2������������������Ӧ������������ˮ�����Fe2O3+3H2SO4=Fe2��SO4��3+3H2O
��������������Ʒ�Ӧ��������ƺ�ˮ�����2HNO3+Ca��OH��2=Ca��NO3��2+2H2O

��ϰ��ϵ�д�
�����Ŀ