��Ŀ����
11��ˮ������֮Դ��������������ϵ���У�����ˮ�������䰮ˮ��Դ����ÿ������Ӧ�������κ�������1��Ϊ�˷�ֹˮ����Ⱦ���������������ڱ���ˮ��Դ���Ǣڢܣ����ţ���
a������ˮ�����ж���ֲ�����������b���������ŷŹ�ҵ��ˮ��
c������ʹ�û���ũҩ����������d��������ˮ�����������������ŷţ�
��Ȼˮ�к����������ʣ�����ˮ���������У����ù��˵ķ�����ȥˮ�е��������ʣ�ͬʱ���������������ClO2��һ�����͵�����ˮ����������ҵ����Cl2��NaClO2��ȡClO2..����ƽ���л�ѧ����ʽ��
1Cl2+2NaClO2�T2NaCl+2ClO2��
��2���ڻ�ѧʵ��Ϳ�ѧ�����У�ˮҲ��һ�ֳ��õ��Լ���ˮ�������ض����������õ�һ��H+���γ�ˮ�������ӣ�H3O+�������ж��������̵���������������AD������ţ���
A����Ԫ�ط����˸ı䡡����������������������B��������ɷ����˸ı�
C�����Ļ�ѧ���ʷ����˸ı䡡������D����ˮ���ӱ�ˮ�����ӵĵ����������˸ı�
�о���Ա������֣���һ�������£���ˮʩ��һ�����糡���ڳ��¡���ѹ�£�ˮ���Խ�ɱ�����Ϊ���ȱ��������ȱ���������ʵ�ü�ֵ���翪����ҩ���������״�ӡ���ȣ�����������CD����д��ţ�
A������ɱ�������B����ֹ�����ۻ�����������C������������������������D���������������
��3��ҽҩ�Ͽ���ʯ�����̶����۲�λ��ʯ������ʯ�ࣨCaSO4•$\frac{1}{2}$H2O��һ�ְ�ɫ��ĩ������ʯ�ࣨCaSO4•2H2O��һ�ּ�Ӳ�Ĺ��壩���֣��̶����۵�ʯ����CaSO4•$\frac{1}{2}$H2O����2CaSO4•H2O�����ѧʽ�����̶�ʱ������Ӧ�Ļ�ѧ����ʽΪ2��CaSO4•$\frac{1}{2}$H2O��+3H2O=2��CaSO4•2H2O����
���� ��1�����Ը��ݺ�������ˮ��Դ�ͷ�ֹ������Ⱦ������з������жϣ��Ӷ��ó���ȷ�Ľ��ۣ����ݻ�ѧ��Ӧǰ��ԭ�ӵ�������������������ƽ��
��2������H+��ˮ����γ�ˮ�������ӵ�ʵ����������
����������Ϣ��ˮʩ��һ�����糡���ڳ��¡���ѹ�£�ˮ���Խ�ɱ�����Ϊ���ȱ�������֪�ȱ��ܽ���̻����ȣ����б����������ʵȣ�
��3��������ʯ�����ʯ������������Լ�����֮����ת����ϵ���ش�
��� �⣺��1������ˮ��Դ�Ĵ�ʩ�����ᵼ��ˮ��Ⱦ������������ʹ�û���ũҩ�����ڱ���ˮ��Դ������ˮ�����ж���ֲ���������ʹˮ�ʱ仵���Ӷ�ʹˮ����Ⱦ����ѡ�ڢܣ����ݻ�ѧ��Ӧǰ��ԭ�ӵ���������������ԭ�����ù۲취���ԶԻ�ѧ����ʽ������ƽ����ϵ��Ӧ�ֱ�Ϊ��1��2��2��
��2��H+��ˮ����γ�ˮ�������ӵķ���ʽΪ��H++H2O=H3O+�����������غ㶨�ɿ�֪��Ԫ������û�б仯��Ԫ�ػ��ϼ�Ҳû�б仯��������ɷ��Ӻ����ӽ�ϳ�Ϊ���Ӷ������仯����ѧ����Ҳ��Ϊ���ԣ�������ǰ�����������Ϊ10���ӣ��ޱ仯����ѡ��A��D��
�����ȱ��ĸ�����γ�������֪���ȱ�ʵ����Ȼ��ˮ���ʲ���������ɱ��ͷ�ֹ�����ۻ�����;�������ȱ����ص㡰�ڳ��¡���ѹ�£�ˮ���Խ�ɱ�����֪�������¿������������ܽ�������������ʴ�Ϊ��C��D��
��3��ҽҩ�Ͽ�����ʯ�����̶����۲�λ����ʯ����һ�����¶��º�ˮ��ϱ�Ϊ�ϴ�Ӳ�ȵ���ʯ�࣬�Ӷ��̶����ã�����ʽΪ��2��CaSO4•$\frac{1}{2}$H2O��+3H2O=2��CaSO4•2H2O����
�ʴ�Ϊ����1���ڢܣ�1��2��2����2��AD��CD����3��CaSO4•$\frac{1}{2}$H2O����2CaSO4•H2O����2��CaSO4•$\frac{1}{2}$H2O��+3H2O=2��CaSO4•2H2O
���� ������һ���ۺ��Ժ�ǿ����Ŀ��Ҫ��ѧ���Ӹ����Ƕ����ջ���֪ʶ���ѶȽϴ���Χ�ơ�ˮ����Ϊ���������龰���Ƚ����������뻯ѧ֪ʶ��ϵ������ͻ���˻�ѧSTSE�������ּ������������֪ʶ����������������ѧ��ʵ��Ӧ��֪ʶ���������Ⲣ���ѣ���Ҫ��ѧ������ȫ�沢�����ף��ر���ԡ��ȱ�������µĸ��Ҫ����ץסʵ����Ȼ��ˮ���ʲ���������ɱ��ͷ�ֹ�����ۻ�����;��

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�| A�� | ϡ���� | B�� | �������Һ | C�� | ̼������Һ | D�� | �Ȼ�����Һ |
| A�� | �÷�Ӧ�����кͷ�Ӧ | B�� | Y��һ��������Ԫ�� | ||
| C�� | Xһ������ʯ�� | D�� | X�������ӻ��ϼ���-2�� |
| A�� | Ӧ�ܷⱣ�� | B�� | ��ȫȼ�յķ�Ӧ�������û���Ӧ | ||
| C�� | ������ȼ�� | D�� | ����N2��H2O��������Ϊ7��9 |
| A�� | ȼ���̻�����ʱ��Զ����Ⱥ�Ϳ�ȼ�� | |
| B�� | ��Ȼ��й©�������رշ��ſ���ͨ�� | |
| C�� | ����ʹ�õļ��õ����Ż�������ˮ���� | |
| D�� | �ֳ�ȼ�ŵ��������ɽ��������������Ϩ�������˳� |
| A�� | �������� | B�� | �ǽ������� | C�� | ������ | D�� | ����� |
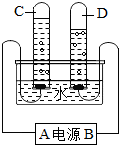 ��ͼΪijͬѧ���е��ˮʵ���װ��ͼ���ݴ˻ش�
��ͼΪijͬѧ���е��ˮʵ���װ��ͼ���ݴ˻ش�