��Ŀ����
ʵ��������һ�ֺ��д��С�մ�Ĺ����ĩ��Ʒ��Ϊ�˲ⶨ����С�մ�ĺ�����ij��ѧ��ȤС���ͬѧ����ʦ��ָ���£���������ͼ��ʾװ�ã����̶ֹ�װ������ȥ������ʵ�顣���������£������ȶ����� �⣬С�մ��ֽ⣬��Ӧ�Ļ�ѧ����ʽΪ��2NaHCO3
�⣬С�մ��ֽ⣬��Ӧ�Ļ�ѧ����ʽΪ��2NaHCO3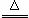 Na2CO3 + CO2�� + H2O ����ʯ����CaO��NaOH�Ĺ��������֪����װ�����������ã�A��C��D����װҩƷ����������ȫ����������ʡ�̽���������£�
Na2CO3 + CO2�� + H2O ����ʯ����CaO��NaOH�Ĺ��������֪����װ�����������ã�A��C��D����װҩƷ����������ȫ����������ʡ�̽���������£�
��. ������Ʒ����װ�����ҩƷ��C��D�������ֱ�Ϊ��m��m C1��m D1��
��. ������Ϊm����Ʒװ��B�������У���ͼ���Ӻ�װ�ã����в���X.
��.�رյ��ɼ�a��b�����ɼ�c���������ܼ��ȡ�
��. ������������Ʒ��ȫ��Ӧ�����ɼ�aͨ���������Ϩ��ƾ��ƣ�ֱ����������ȴ��
��. �ٴγ���C��D���������ֱ�Ϊ m C2��m D2��
��ش��������⣺
��1��������в���X����Ϊ ��
��2��C����װҩƷΪ_____________��
��3��D�з�����Ӧ�Ļ�ѧ����ʽΪ_____________ ��
��4����������ɼ�aͨ���������Ϩ��ƾ��ƣ�ֱ����������ȴ�Ķ��������� ��
��5������ʵ��ʵ�ʣ�ѡ��ʵ���������ݣ�����Ʒ��С�մ����ı���ʽΪ ��
�رյ��ɼ�c�����ɼ�a��b����ͨ�����
Ũ����
2NaOH+CO2===Na2CO3+H2O
��������CO2��ˮ�������������������𣲵㣩
42��mD2- mD1����11 m

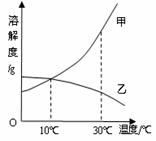
�±���KNO3�IJ����ܽ�����ݣ���ش����⣺
��1��40��ʱ����70gKNO3�������100gˮ�У���ֽ��裬�γɵ���Һ����Ϊ����g��
��2��60��ʱ��KNO3�ı�����Һ�еõ������һ�ַ�����������
��3�����ݱ������ݣ�������ͼ����ͼ�л���KNO3�ܽ�����ߵIJ�ͼ��
| �¶�/�� | 0 | 20 | 40 | 60 |
| �ܽ��/g | 13.3 | 31.6 | 63.9 | 110 |
�����й��������������˵����ȷ����
| �� | A�� | �л�����顢�Ҵ��������� |
| �� | B�� | ������Ⱦ�������̼���������������� |
| �� | C�� | ����ú��ʯ�͡���ˮ������ |
| �� | D�� | �ϳɲ��ϣ��Ͻ𡢺ϳ����ϳ���ά |
 N2 + 3Cu + 3H2O ��ij��ѧ��ȤС��ͬѧ�����ͼ��ʾʵ������֤��������ɣ�ͼ�мг֡��̶�װ�ò��־���ȥ������֪����װ�����������ã���ʵ���������Լ���������B��D��E����װҩƷ����ȫ����������ʡ�ʵ��ǰװ�� C��D �������ֱ�Ϊ mC1��mD1 .
N2 + 3Cu + 3H2O ��ij��ѧ��ȤС��ͬѧ�����ͼ��ʾʵ������֤��������ɣ�ͼ�мг֡��̶�װ�ò��־���ȥ������֪����װ�����������ã���ʵ���������Լ���������B��D��E����װҩƷ����ȫ����������ʡ�ʵ��ǰװ�� C��D �������ֱ�Ϊ mC1��mD1 .
 ʾ��~��װ�ã��ɹ�ʵ������ȡ�й�����ʱѡ�á�
ʾ��~��װ�ã��ɹ�ʵ������ȡ�й�����ʱѡ�á�
 �����Dz�������Һ��
�����Dz�������Һ��