��Ŀ����
11��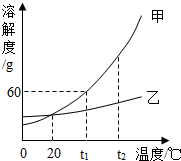 ������ճ������ũҵ�����벻��ˮ����ش�
������ճ������ũҵ�����벻��ˮ����ش���1��������ˮ��Ӳˮ�ķ�����ȡˮ�����Թ��У����Թ��м������ˮ����������ĭ�϶࣬������ˮ����������ĭ���١��д�����������Ӳˮ��
��2������ˮ�ķ����У���Ծ����̶Ƚϸߵ�������
��3��ˮ��ͨ��������¿��Էֽ⣬д���÷�Ӧ�Ļ�ѧ����ʽ2H2O$\frac{\underline{\;ͨ��\;}}{\;}$2H2��+O2����
��4���������ֹ������ʵ��ܽ��������ͼ��ʾ���ش��������⣺
��t1��ʱ����60g�����ʷ���100gˮ�У���ֽ��裬������Һ��������������w���ף���ͬ���������ʵı�����Һ��������������w���ң���С��ϵΪw���ף���w���ң����������������=����
�������к����������ң���ȥ�ҵķ�������ȴ�ȱ�����Һʹ�ᾧ������
���� ��1������Ӳˮ����ˮ�ļ��鷽�������з������
��2�����ݾ���ˮ�ķ����У���Ծ����̶Ƚϸߵ���������з�����
��3������ˮͨ��ֽ������������������н��
��4�������ܽ�ȵĸ�������������ʵ��ܽ�����߷������
��� �⣺��1��Ӳˮ����ˮ���������������ĸ�þ���ӵĶ��٣����÷���ˮ������Ӳˮ����ˮ��ȡˮ�����Թ��У����Թ��м������ˮ����������ĭ�϶࣬������ˮ����������ĭ���١��д�����������Ӳˮ��
��2������ˮ�ķ����У���Ծ����̶Ƚϸߵ�������
��3��ˮͨ��ֽ�������������������Ӧ�Ļ�ѧ����ʽΪ��2H2O$\frac{\underline{\;ͨ��\;}}{\;}$2H2��+O2����
��4�����ɼ����������ʵ��ܽ�����߿�֪��t1��ʱ�����ܽ�ȴ����ҵ��ܽ�ȣ�����t1��ʱ���ܽ����40g���ɴ˿�֪��t1��ʱ���ױ�����Һ�����������������������ʱ�����Һ��������������������Ϊ���ܽ�����¶�Ӱ��ϴ������¶�Ӱ���С�����������к����������ң���ȥ�ҿ�����ȴ�ȱ�����Һʹ�ᾧ�����ķ�����
�ʴ�Ϊ��
��1��ȡˮ�����Թ��У����Թ��м������ˮ����������ĭ�϶࣬������ˮ����������ĭ���١��д�����������Ӳˮ����2������3��2H2O$\frac{\underline{\;ͨ��\;}}{\;}$2H2��+O2������4���٣�������ȴ�ȱ�����Һʹ�ᾧ�������������ɣ���
���� ���⿼����Ӳˮ������������ˮ�ķ�����ѧ����ʽ����д���ܽ�ȵĸ���ȣ�������ǿ��

 ϰ�⾫ѡϵ�д�
ϰ�⾫ѡϵ�д�| A�� | 3H2O�еġ�3����ʾ3��ˮ���� | |
| B�� | Al3+�С�3����ʾ��Ԫ�صĻ��ϼ�Ϊ+3�� | |
| C�� | SO3�еġ�3����ʾһ��������������к���3����ԭ�� | |
| D�� | 3C�еġ�3����ʾ3��̼ԭ�� |
| A�� | �����������ͬ | B�� | ������������ͬ | ||
| C�� | ������������ͬ | D�� | ������������ͬ |
��ʾʵ�齫һ������ϡH2SO4���뵽ʢ��NaOH��Һ��С�ձ��У�
��ѧ�����ݡ��÷�Ӧ�Ļ�ѧ����ʽH2SO4+2NaOH�TNa2SO4 +2H2O��
��������⡿ʵ����δ�۲쵽����������ͬѧ�������ʣ���Ӧ����Һ��������ʲô�أ�
���������������ʣ���ҷ�������룮����ͬѧ�IJ������£�
����һ��ֻ��Na2SO4 ���������Na2SO4��H2SO4
����������Na2SO4��NaOH �����ģ���Na2SO4��H2SO4��NaOH
����ͬѧ�����ϲ���������ɣ���Ϊ��һ�ֲ����Dz������ģ��������IJ����ǣ�
��ʵ��̽����
��1������ͬѧȡ����С�ձ��е���Һ�������Թ��У��μӼ���Fe2��SO4��3��Һ�������Ա仯����Һ��һ��û�У�
��2��Ϊ����֤������룬��ѧϰС�������ձ��е���Һ����ѡ����ʦ�ṩ��pH��ֽ��ͭƬ��BaC12��Һ��Na2CO3��Һ��������������������̽��������дʵ������
| ʵ�鷽�� | ����ҺpH | �μ�Na2CO3��Һ | �μ�BaC12��Һ |
| ʵ����� | 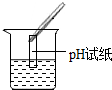 | 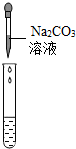 | 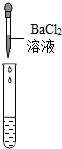 |
| ʵ������ | pH��7 | ������ | ���ɰ�ɫ���� |
| ʵ����� | ��Һ����H2SO4 | ��Һ����H2SO4 | ��Һ����H2SO4 |
�����۷�˼����ʦ��ͬѧ�����ö��ַ�������̽�������ҵó���ȷ���۸���϶���ͬʱָ��������ʵ��̽��
��2���д����������Դ���
ʵ�鷽���еĴ�����BaC12��Na2SO4��ӦҲ�����ɰ�ɫ����������֤��һ����H2SO4��
ʵ������еĴ�����pH��ֽ������Һ�У�
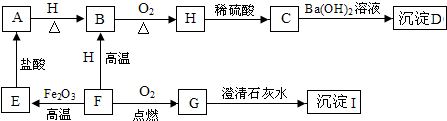
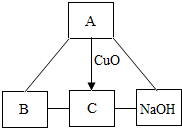 A��B��C�dz��л�ѧ�����IJ�ͬ���͵����ʣ�����֮������ͼת����ϵ����-����ʾ�����������Ӧ����������ʾ�ɷ�Ӧ��ת���������A������θҺ�к��е��ᣬBΪ������������Ľ������ش��������⣺
A��B��C�dz��л�ѧ�����IJ�ͬ���͵����ʣ�����֮������ͼת����ϵ����-����ʾ�����������Ӧ����������ʾ�ɷ�Ӧ��ת���������A������θҺ�к��е��ᣬBΪ������������Ľ������ش��������⣺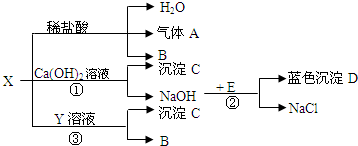
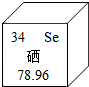 ��������������Ԫ�أ���ͼ������Ԫ�����ڱ��е���Ϣ����ͼ��֪�������ԭ������Ϊ78.96����ԭ�ӵ�������Ϊ34��
��������������Ԫ�أ���ͼ������Ԫ�����ڱ��е���Ϣ����ͼ��֪�������ԭ������Ϊ78.96����ԭ�ӵ�������Ϊ34��