��Ŀ����
����������������벻��������
��1��Ŀǰ�������ߵĽ����� ��
��2�����ڳ�ʪ�Ŀ�����ᷢ����ʴ��֤������һ���μ��˷�Ӧ����Ҫ����ʵ���� ��
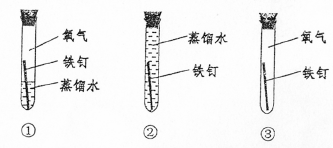
A���٢ڡ��������� B���٢� C���ڢ� D���٢ڢ�
(3) Fe2O3����Ԫ�ص���������Ϊ____________��ij����������Ҫ�ɷ���(Fe2O3���ij�����ʯ������������ÿ����Ҫ����1000t������20%�ij�����ʯ���ó������Ͽɲ���Fe98%�� ������������ ��
������������ ��
��4��������������ˮ�����������ǿ����Һ������������������Һ����Ӧ����ƫ�����ƣ�NaAlO2����ˮ������������Ϣд��������������������Һ��Ӧ�Ļ�ѧ����ʽ�� ��
��5��������ͭ������п��ϡ����Ļ����Һ�У�������������ۣ�������ʣ�ࣩ��ʹ֮��ַ�Ӧ���ˣ��������ijɷ��� ����Һ�к��е����� ��
(1) Fe (1��) �� (2) A (1��) ; (3) 70% (1��) ; 571.4 t(2��)
(4) Al2O3��2NaOH == 2NaAlO2��2H2O (2�֡���ѧʽ����Ϊ0�֣�û����ƽ��1��)
��5��Fe��Cu(2��)�� Zn2+ ��Fe2+��SO42- ��H2O (3�֡�)

 ��˼ά������ҵ���ټ��ִ�ѧ������ϵ�д�
��˼ά������ҵ���ټ��ִ�ѧ������ϵ�д�