��Ŀ����
��2011?��ƽ����ģ���ں�ۡ��ۺͷ���֮�佨����ϵ�ǻ�ѧѧ�Ƶ��ص㣮��1�������Ǽ������ӵĽṹʾ��ͼ��

���DZ�ʾ��Ԫ�أ���ʾ����ԭ�ӣ���ʾ���������ӣ�����ţ���
��2��A���ʳ����ں��Ӻ��и��������Է�������Ϊ26����һ�������Ĵ�����A��40g�Ĵ�����B��һ�������°���ͼ��ʾ��ַ�Ӧ����B���ʷ�Ӧ��ȫʱ��������44gC���ʺ�9gD���ʣ�

�ٸ÷�Ӧ�Ļ�ѧ����ʽΪ��
��A�����и�Ԫ�ص�������Ϊ��
���𰸡���������1��Ȧ�ڱ�ʾ������������Ԫ���������������������ģ�ԭ�������������ڵ��������������Ǻ������������������������
��2�����������غ㶨�ɣ���Ӧǰ����A��������μӷ�Ӧ��B���ʵ�����40g�������͵���������C������44g��D������9g�����ͣ��ɴ˿ɼ���μӷ�Ӧ������A��������
������B��C��D�ķ�����ͼ��֪����BΪ����������CΪ������̼������DΪˮ�����ݷ�Ӧǰ��Ԫ�����ࡢ�������䣬��44gC�����е�CԪ�ء�9gD����ˮ�е�HԪ��ȫ������������A��������A��C��HԪ�ص���������μӷ�Ӧ����A��������ϵ�����ж�����A����ɣ����ɶ�����̼��ˮ���������������A�����Ԫ�ص������ȣ�����ȷ���û�ѧʽ��
����⣺��1���нṹʾ��ͼ��֪��Ȧ�ڱ�ʾ������������Ԫ���������������������ģ�ԭ�������������ڵ��������������Ǻ������������������������
�ʴ�Ϊ�������٢ۢܣ��ڣ�
��2��һ�������Ĵ�����A��40g�Ĵ�����B��һ�������³�ַ�Ӧ����B���ʷ�Ӧ��ʱ��������44g C���ʺ�9gD���ʣ����������غ㶨�ɣ���μӷ�ӦA���ʵ�����=44g+9g-40g=13g��
���ݷ�Ӧ����ʾ��ͼ��֪���÷�ӦΪ����A��B����������Ӧ����C���ʶ�����̼��D����ˮ�������������̼�е�CԪ�ء�ˮ��HԪ��ȫ������������A��������A��CԪ�ص�����=44g×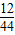 ×100%=12g��HԪ������=9g×
×100%=12g��HԪ������=9g× ×100%=1g������C��HԪ��������=12g+1g=13g���õ��ڲμӷ�Ӧ��A���ʵ�������˵������A��ֻ��C��H����Ԫ�أ���C��H��Ԫ�ص�������=12g��1g=12��1����̼��ԭ�Ӹ�������1��1�����Ǻ��ӳ������壬���������C2H2��
×100%=1g������C��HԪ��������=12g+1g=13g���õ��ڲμӷ�Ӧ��A���ʵ�������˵������A��ֻ��C��H����Ԫ�أ���C��H��Ԫ�ص�������=12g��1g=12��1����̼��ԭ�Ӹ�������1��1�����Ǻ��ӳ������壬���������C2H2��
�ʴ�Ϊ����2C2H2+5O2 4CO2+2H2O��12��1��
4CO2+2H2O��12��1��
�������������۽ṹʾ��ͼ�Ŀ��飬����ʱ��ԭ�ӽṹʾ��ͼ��ʾ��������˽⣬�Լ��������ʵĻ�ѧʽ��һ�������Ļ�������ij�����Ԫ�ص�����=�����������×�������и�Ԫ�ص������������ǽ���Ĺؼ����ڣ�
��2�����������غ㶨�ɣ���Ӧǰ����A��������μӷ�Ӧ��B���ʵ�����40g�������͵���������C������44g��D������9g�����ͣ��ɴ˿ɼ���μӷ�Ӧ������A��������
������B��C��D�ķ�����ͼ��֪����BΪ����������CΪ������̼������DΪˮ�����ݷ�Ӧǰ��Ԫ�����ࡢ�������䣬��44gC�����е�CԪ�ء�9gD����ˮ�е�HԪ��ȫ������������A��������A��C��HԪ�ص���������μӷ�Ӧ����A��������ϵ�����ж�����A����ɣ����ɶ�����̼��ˮ���������������A�����Ԫ�ص������ȣ�����ȷ���û�ѧʽ��
����⣺��1���нṹʾ��ͼ��֪��Ȧ�ڱ�ʾ������������Ԫ���������������������ģ�ԭ�������������ڵ��������������Ǻ������������������������
�ʴ�Ϊ�������٢ۢܣ��ڣ�
��2��һ�������Ĵ�����A��40g�Ĵ�����B��һ�������³�ַ�Ӧ����B���ʷ�Ӧ��ʱ��������44g C���ʺ�9gD���ʣ����������غ㶨�ɣ���μӷ�ӦA���ʵ�����=44g+9g-40g=13g��
���ݷ�Ӧ����ʾ��ͼ��֪���÷�ӦΪ����A��B����������Ӧ����C���ʶ�����̼��D����ˮ�������������̼�е�CԪ�ء�ˮ��HԪ��ȫ������������A��������A��CԪ�ص�����=44g×
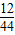 ×100%=12g��HԪ������=9g×
×100%=12g��HԪ������=9g× ×100%=1g������C��HԪ��������=12g+1g=13g���õ��ڲμӷ�Ӧ��A���ʵ�������˵������A��ֻ��C��H����Ԫ�أ���C��H��Ԫ�ص�������=12g��1g=12��1����̼��ԭ�Ӹ�������1��1�����Ǻ��ӳ������壬���������C2H2��
×100%=1g������C��HԪ��������=12g+1g=13g���õ��ڲμӷ�Ӧ��A���ʵ�������˵������A��ֻ��C��H����Ԫ�أ���C��H��Ԫ�ص�������=12g��1g=12��1����̼��ԭ�Ӹ�������1��1�����Ǻ��ӳ������壬���������C2H2���ʴ�Ϊ����2C2H2+5O2
 4CO2+2H2O��12��1��
4CO2+2H2O��12��1���������������۽ṹʾ��ͼ�Ŀ��飬����ʱ��ԭ�ӽṹʾ��ͼ��ʾ��������˽⣬�Լ��������ʵĻ�ѧʽ��һ�������Ļ�������ij�����Ԫ�ص�����=�����������×�������и�Ԫ�ص������������ǽ���Ĺؼ����ڣ�

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
��2011?��ƽ����ģ��ʵ������5�ְ�ɫ���壬��ʦ�û�ѧʵ��С��ͬѧ������Щ���壮
��1�������о�
��5�ְ�ɫ����ֱ��ǣ�NaOH��Na2CO3��CuSO4��Ca��OH��2��NaHCO3��
�ڲ������ϣ�С��ͬѧ������NaOH��Na2CO3��NaHCO3��һЩ���ʣ�
��2��ʵ��̽����ͬѧ�ǵ�̽���������£����������ɣ�
��3��ʵ�鷴˼����ʦ����ͬѧ������������ʵ��̽�����̣�����ȫ�淴˼������ʵ��������в�ȷ���ԣ�����ȷ����������______���ѧʽ������������������������
��1�������о�
��5�ְ�ɫ����ֱ��ǣ�NaOH��Na2CO3��CuSO4��Ca��OH��2��NaHCO3��
�ڲ������ϣ�С��ͬѧ������NaOH��Na2CO3��NaHCO3��һЩ���ʣ�
| ���� | NaOH | Na2CO3 | NaHCO3 |
| �ܽ��� | 20��ʱ�ܽ��109g | 20��ʱ�ܽ��21.5g �ܽ�ʱ���� | 20��ʱ�ܽ��9.6g �ܽ�ʱ���� |
| ���� | ���ֽ� | ���ֽ� | �����ֽⷴӦ���ֽ����Ϊ̼���ơ�������̼��ˮ�� |
| ʵ�鲽�� | ʵ������ | ʵ����� | |
| ��һ������5֧�Թ��зֱ����������5�ְ�ɫ���壬 �ֱ����A��B��C��D��E��Ȼ�����μ���һ������ˮ���� | A��C��D����ҺΪ��ɫ��B����ҺΪ���ǣ�E����Һ����ɫ�� | B��______ E��______ | |
| �ڶ�����������A��C��D��Һ�зֱ�μ���ɫ�ķ�̪��Һ�� | ��֧�Թ��еķ�̪��Һ����죮 | ���ۣ� ______ | |
| ��������������ͼװ�ã���A��B��C����ʵ�飮 |  | ||
| �ټ��ȹ���A | B��Һ���������� | A��Na2CO3 | |
| �ڴ���ֹͣ���Ⱥ�ͨ������©����C��Һ�м���ϡ���� | C��Һ�������ݲ�����B��Һ���������ݳ����а�ɫ�������ɣ� | B��Һ�з�����Ӧ�Ļ�ѧ����ʽΪ______ | |
| ���IJ���������ͼװ�ã���B��C����ʵ�飮 |  | �����ȵ��Թܿ�����ɫҺ�����ɣ�B��Һ���������ݳ�����Һ����ǣ� | C��______ �����ų����ó��� D��NaOH |
| ʵ�鲽�� | ʵ������ͽ��� |
| ______ | ______ |


