��Ŀ����
11�����ž��õķ�չ����Դ�ͻ��������Ϊ���ǹ�ע�Ľ��㣮��1��ú��ȼ��ʱ����ú������ú����Ϊ��ʹú�������ֽӴ���
��2���Դ���������ֹĦ�г��Ժܶ����������г�����Ч�ؼ����˶�����̼����������һ����̼��������ŷţ���Щ�����л���������ЧӦ���Ƕ�����̼�����������Ƕ�������
��3����̼������ָ������Ҫ���������������ĺͲ������ģ��Ӷ����Ͷ�����̼���ŷ����������������ϵ�̼�����������AB������ĸ��ţ���
A������һ���Ե�ľ�� B�����������ε���ͷC��������չ�������磮
���� ��1���ٽ���ȼ��ȼ�յķ����У������ȼ���������ĽӴ����������������Ũ�ȣ��ݴ˽��з������
��2��������������ЧӦ�������ԭ���з������
��3������̼������ָ����������Ϣʱ�����õ�����Ҫ�������٣��ر��Ǽ��ٶ�����̼���ŷ�����������̬�����Դӽڵ硢���ܺͻ��յȻ������ı�����ϸ�ڣ��ݴ˽��з�����ɣ�
��� �⣺��1��ú��ȼ��ʱ����ú������ú����Ϊ��ʹú�������ֽӴ�������ú���ȼ�գ�
��2���ܶ����������г�����Ч�ؼ����˶�����̼����������һ����̼��������ŷţ���Щ�����л���������ЧӦ���Ƕ�����̼�����������Ƕ�������
��3��A������һ���Ե�ľ�꣬�ܽ�Լ��ֽ���õ�ľ�ģ�������ľ�Ŀ�������ǿ������̼�����ģ���ѡ����ϡ���̼������
B�����������ε���ͷ���ܽ�Լ���ܣ��ܼ��ٶ�����̼���ŷţ���ѡ����ϡ���̼������
C��������չ�������磬����������Ķ�����̼����ѡ����ϡ���̼������
�ʴ�Ϊ����1����ֽӴ�����2��������̼����������3��AB��
���� �����ѶȲ������մٽ���ȼ��ȼ�յķ���������ЧӦ�����ꡢ��̼�������������ȷ�����Ĺؼ���

| A�� | �٢ڢۢܢݢ� | B�� | �ܢ٢ۢڢݢ� | C�� | �٢ۢڢܢݢ� | D�� | �٢ܢڢۢݢ� |
| ����������� | ����ķ��� | |
| A | ��ë����� | ȼ�գ�����ζ |
| B | �ռ�������� | ��ˮ������Һ����¶ȱ仯 |
| C | �������Ͻ� | ��̻� |
| D | �Ȼ��������� | ����ʯ�ң���ĥ |
| A�� | A | B�� | B | C�� | C | D�� | D |

��֪������ʵ��ܽ�ȣ�20�棩�����
| ���� | K2CO3 | K2SO4 | KMnO4 |
| �ܽ��/g | 111 | 11.1 | 6.34 |
��2��������������¶Ȳ��˹��ߵ�ԭ���Ǹ�����������ֽ⣮
��3����ͨCO2�ữ������Ӧ�Ļ�ѧ����ʽΪ��3K2MnO4+2CO2�T2KMnO4+MnO2��+2K2CO3����ϡH2SO4�ữ����Ӧ�Ļ�ѧ����ʽΪ��3K2MnO4+2H2SO4�T2KMnO4+MnO2��+2K2SO4+2H2O��ҵ�ϲ�����ϡH2SO4�ữ��ԭ������ͬ�����£�����غ�����ص��ܽ�ȶ��Ƚ�С��������Ũ���ᾧ�ķ������Է��룮
��4����ⷨҲ����ʵ����K2MnO4��KMnO4��ת������Ӧ�Ļ�ѧ����ʽΪ��3K2MnO4+2H2O$\frac{\underline{\;ͨ��\;}}{\;}$2KMnO4+2KOH+H2���롰ͨCO2�ữ����ȣ����ŵ��ǣ����һ�㼴�ɣ����ɵ��������ؿ���ѭ��ʹ�ã�
| A�� | ʯī�ͽ��ʯ������̼�ĵ��� | |
| B�� | CO2��ʹ�������ɫʯ��ֽ����� | |
| C�� | CO ���CO2�� | |
| D�� | ľ̿��CO ��һ�������¶���������ͭ��Ӧ |
 ��Һ����Һ�ڻ�ѧ�о���ʵ������������Ҫ���ã�
��Һ����Һ�ڻ�ѧ�о���ʵ������������Ҫ���ã�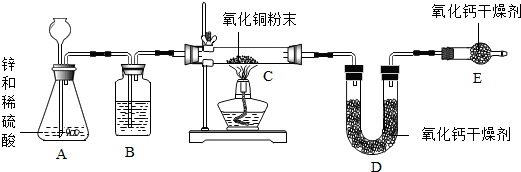
 ��ͼ��a��b��c�������ʵ��ܽ�����ߣ�����ͼ�ش�
��ͼ��a��b��c�������ʵ��ܽ�����ߣ�����ͼ�ش�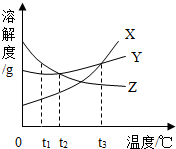 ��ͼ��X��Y��Z���ֹ������ʵ��ܽ������ͼ����������ͼ�ش��������⣮
��ͼ��X��Y��Z���ֹ������ʵ��ܽ������ͼ����������ͼ�ش��������⣮