��Ŀ����
��ͼ��ʵ������ȡ�����һЩ����װ�á���ش��������⣺
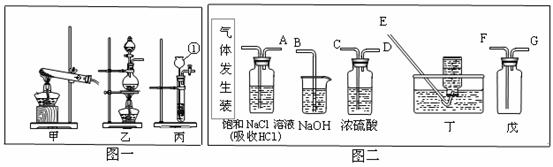
��1������������������� ��
��2��ʵ�����ø��������ȡ��������Ӧ�Ļ�ѧ����ʽΪ ��Ӧ��ѡ��ķ���װ��Ϊ ���ö�װ���ռ�����ʱ���ж��������ռ����������� ��
��3����ȡ������̼ʱ���ñ�װ��������װ�ã�ʵ�����������Ҫ��ͣ��Ӧ�ò�ȡ�Ĵ�ʩ��
��
��4��������Cl2��ͨ�������Ϊ�д̼�����ζ�Ļ���ɫ���ж����壬�ܶȱȿ����ɸ�ˮ��Ӧ����Ӧ����Һ�����ԡ�ʵ����ͨ����Ũ������������̹��ȵķ�������ȡ������ʵ������ȡ�����ķ���װ��Ϊ ����ͼ���е���������һ��ȫ���õ�����������ľ���������ռ���β������������ȷ�����ӷ����ǣ�A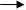
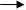
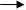 B��������ĸ��
B��������ĸ��
��1������©����1�֣�
��2��2KMnO4  K2MnO4 + MnO2 + O2����2�֣� �ף�1�֣�
K2MnO4 + MnO2 + O2����2�֣� �ף�1�֣�
���ݴӼ���ƿƿ������ð��1�֣�
��3���رյ����ϵĻ�����1�֣�
��4���ң�1�֣� C D F G ��2�֣�ȫ�ԲŸ��֣�

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
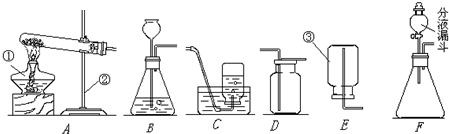


 ��ͼ��ʵ������ȡ����ʱ���õ�װ�ã���ش��������⣮
��ͼ��ʵ������ȡ����ʱ���õ�װ�ã���ش��������⣮
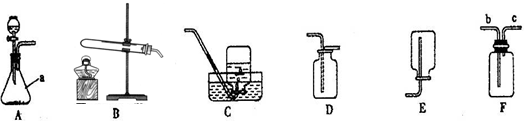
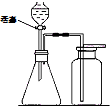 ������ɣ��ȹرջ��������ڼ���ƿ�м�ˮ��û����ĩ�ˣ�Ȼ��
������ɣ��ȹرջ��������ڼ���ƿ�м�ˮ��û����ĩ�ˣ�Ȼ��