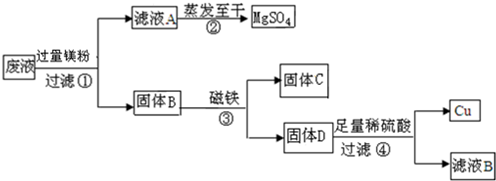
��Ŀ����
18����ͭ����п��ͭ�γɵĺϽ��н�ǿ����ĥ���ܣ����������й㷺����;����λͬѧΪ�˲ⶨ��ͭм��Ʒ��ɣ�������ȡ��Ʒ��ϡ���ᷴӦ����ʵ�����ݼ�¼�����| �� | �� | �� | |
| ��Ʒ����/g | 25 | 30 | 25 |
| ϡ��������/g | 120 | 80 | 80 |
| ��ַ�Ӧ��ʣ����/g | 144.8 | 109.8 | 104.8 |
��1�����������غ㶨�ɣ�������ס��ҡ�����λͬѧ������0.2g������ͨ��������λͬѧ��ʵ�����ݽ��������Աȣ��ɵó���ͬѧ��ȡ��ϡ�������ͭ����ܷ���ǡ����ȫ��Ӧ��
��2�������ͬѧ��ȡ��ϡ�������ͭǡ����ȫ��Ӧ���������
������ȡ�Ļ�ͭ��Ʒ��п������������
��������Һ��������������������������ȷ��0.1%��
���� ��1���ɱ������ݷ�����֪��ͬѧ�ͱ�ͬѧ��ʵ���������������ͬ����Ʒ������ͬ������ͬѧ�õ�ϡ���������࣬˵����ͬѧ��ʵ��ϡ�����������ͬѧ�ͱ�ͬѧ��ʵ����ϡ����������ͬ����Ʒ������ͬѧ�Ķ࣬������������������ͬ��˵����ͬѧ��ʵ����Ʒ���������Ϸ�������ͬѧ��ʵ������Ʒ��ϡ����ǡ����ȫ��Ӧ��
��2�������������������������п������������п������������Ʒ���������ٷ�֮�ټ��ɣ�
��3�����������������������������п������������������Һ������п������������
��� �⣺��1�����������غ㶨�ɣ�������ס��ҡ�����λͬѧ������0.2g������
�ɱ������ݷ�����֪��ͬѧ�ͱ�ͬѧ��ʵ���������������ͬ����Ʒ������ͬ������ͬѧ�õ�ϡ���������࣬˵����ͬѧ��ʵ��ϡ�����������ͬѧ�ͱ�ͬѧ��ʵ����ϡ����������ͬ����Ʒ������ͬѧ�Ķ࣬������������������ͬ��˵����ͬѧ��ʵ����Ʒ���������Ϸ�������ͬѧ��ʵ������Ʒ��ϡ����ǡ����ȫ��Ӧ��
��2������Ʒ��Zn������Ϊx������ZnSO4������Ϊy
Zn+H2SO4�TZnSO4+H2��
65 161 2
x y 0.2g
$\frac{65}{x}=\frac{2}{0.2g}$
x=6.5 g
��ͭ��Ʒ��п����������Ϊ��$\frac{6.5g}{25g}��100%=26%$
��3��$\frac{161}{2}=\frac{y}{0.2g}$
y=16.1 g
������Һ��������������Ϊ��$\frac{16.1g}{6.5g+80g-0.2g}��100%=18.7%$
��$\frac{16.1g}{16.1g+��80g-9.8g��}��100%=18.7%$��$\frac{16.1g}{104.8g-��25.0g-6.5g��}��100%=18.7%$
�𰸣�
��1��0.2g�� ����
��2��������ȡ�Ļ�ͭ��Ʒ��п����������Ϊ26%��
��������Һ��������������Ϊ18.7%��
���� �����ؼ���Ҫͨ��ͼ�������ݷ����������������1.0g�������÷���ʽ�����п������п���������ٽ�һ�����м��㣮

| A�� | �� | B�� | ���� | C�� | ���� | D�� | ʳ�� |
| A�� | 1mol��O2�к���1.204��1024����ԭ�ӣ��ڱ�״����ռ�е����ԼΪ22.4L | |
| B�� | 1molO3��1.5molO2������ͬ����ԭ���� | |
| C�� | ��CO2��O2��ɵĻ�����й���NA�����ӣ����е���ԭ����2NA | |
| D�� | ��״���£�11.2LH2O�������ķ�����Ϊ0.5NA�� |
| A�� | ̿���տ� | B�� | ��˿��ˢ�� | C�� | ������� | D�� | ��ţ�̱��� |
| A�� | ����Լռ���������$\frac{1}{5}$ | |
| B�� | �����������˿�������� | |
| C�� | ��������Ⱦָ��Խ��������״��Խ�� | |
| D�� | ������ϡ�����塢������̼�������������� |
���ձ������Թܣ����ԹܼУ��ܾƾ��ƣ���ʯ������������̨��
| A�� | �٢ܢ� | B�� | �ڢۢ� | C�� | �ڢۢܢ� | D�� | �٢ܢݢ� |
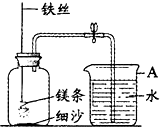 ijУ�о���ѧϰС������ͼװ�ý���þ���ڿ�����ȼ�յ�ʵ�飬ȼ�ա���ȴ���ֹˮ�У����뼯��ƿ��ˮ�����Լռ����ƿ�ݻ���70%��
ijУ�о���ѧϰС������ͼװ�ý���þ���ڿ�����ȼ�յ�ʵ�飬ȼ�ա���ȴ���ֹˮ�У����뼯��ƿ��ˮ�����Լռ����ƿ�ݻ���70%��