��Ŀ����
3��ˮ-----����֮Դ��1���ճ�����������Ӳˮ����ˮ�÷���ˮ�������н�Ӳˮ��������з���
��2������ˮ�����ö�������ɱ��������д���������ȵĻ�ѧʽ�������Ԫ�صĻ��ϼۣ�
��3��ij��ֳ�����ⶨˮ����ˮ�������ǿ������ѡ��C������ţ�
A��ɫʯ����ҺB��̪��ҺCpH ��ֽ
��4��̽��ˮ�����
��ѧ����ͼ1װ �����ⶨ ˮ���� �ɣ����� a Ϊ�� Դ�ĸ� �����������������ʵ��ʱ��������������������ǿˮ�ĵ����ԣ���һ��ʼ������ˮ������Ϊ49.7�ˣ����� 0.3 �˵�����������ȫ�ܽ��ͨ��һ��ʱ�����Һ�е���������������Ϊ0.625%��������ab�ϣ����Թ��п����ռ�������1.8�ˣ���ȷ��С�����һλ������ʵ��õ�����ˮ����ɵĽ�����ˮ������������Ԫ����ɵģ�
��5���ⶨˮ�и�ԭ�ӵĸ�����
ʵ�����ô��������������ԭ����ͭ����ͨ��ʵ����������װ���е������仯�� �ⶨˮ������ԭ�Ӹ����ȣ�ʵ�鲽�����£��� ����ÿ����������ȫ��������һ��ʼװ���� ������ʵ������Ӱ�죩
��֪���ټ�ʯ�����������ƺ������ƵĻ���������ˮ�Ͷ�����̼��
��������ͭ���ȷֽ������ˮ������ͭ ��һ��������װ�ã����װ�����ܣ�
�ڶ������ֱ�Ƶ� C �в����ܺ�ҩƷ������101.6�ˣ�D��װ����ͬҩƷ������120.0��
��������ͨһ��ʱ�����������ʼ�� C �������ܼ��ȣ�
���IJ���ֹͣ���ȣ�����ͨһ��ʱ���������
���岽���ٴγƵ� C �в����ܺ�ҩƷ������100.0�ˣ�D��װ����ͬҩƷ������121.8��
��д���������������a����̨b�ƾ���
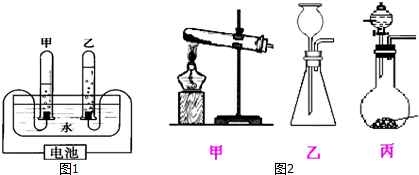
����ͼ3��ʵ������п�ͽ�Ũ�����ᷴӦ���õ������ȶ���������������Ӧ��ѡ�õķ���װ��Ϊͼ2�еļף�ѡ���ס����ҡ����������������������Ļ�ѧ����ʽΪZn+2HCl�TZnCl2+H2��
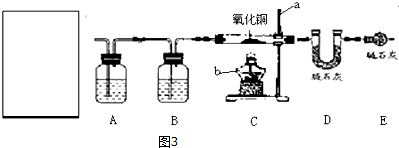
��A��B װ���зŵ�ҩƷ�ֱ�Ϊc������ţ�
a��A �б��͵�̼��������ҺB ��Ũ����
b��A ��Ũ����B �б��͵�̼��������Һ
c��A ��Ũ����������ҺB ��Ũ����
d��A ��Ũ����B ��Ũ����������Һ
��ʵ�鿪ʼͨһ��ʱ���������Ŀ���dz���װ���еĿ�����ȷ�ϴ�Ŀ���Ѿ��ﵽ��ʵ��� ������Ϊ��Eװ��β���ռ����岢�鴿��
�ݼ���һ��ʱ���C �з�����Ӧ�ķ���ʽΪCuO+H2$\frac{\underline{\;\;��\;\;}}{\;}$Cu+H2O��ʵ�������ֹͣ���ȣ�����ͨ���� ��Ŀ��֮һ�������ɵ�ͭ�����ȴ����һĿ��Ϊ�����ɵ�ˮ����ȫ����
��E װ�õ�����Ϊ��ֹ�����е�ˮ�Ͷ�����̼����װ��D������װ�ã��ᵼ��ʵ���õ�����������ƫ���ƫ��ƫС������Ӱ�족��
�����ʵ�鷽����õ�����������Ϊ2��1
��ʵ�鷴˼�������������Ӱ��ʵ�����������ԭ�Ӹ����Ȳⶨ����AB����ţ� A������ͭû��ȫ������ԭB������ͭ�л�������������
C������ͭδ��ȫ����D������ͭ�л�������������ͭ
��������ʵ���У��μӷ�Ӧ������ֻռʵ�����������������֮һ������ɴ�ʵ�������� Ҫ����Ϊ 80%�Ĵ�п���ٿˣ����ʲ����ᷴӦ����
���� ��1�����ݷ���ˮ��������Ӳˮ����ˮ��������п��Խ���ˮ��Ӳ�Ƚ��з�����
��2�����ݻ�������Ԫ�ػ��ϼ۴�����Ϊ����з�����
��3������pH��ֽ���Բⶨ��Һ�����Ƚ��з�����
��4�����ݵ��ˮʱ�������������������������������������������������������ԼΪ1��2���з�����
��5������п��ϡ���ᷴӦ�����Ȼ�п���������з�����
��������������Һ�ܹ������Ȼ������壬Ũ����������������������з�����
���ݼ��������£�����ͭ��������Ӧ����ͭ��ˮ���з�����
�����ṩ�����ݿ��Լ���ˮ������ԭ�Ӹ����ȣ�
�����ṩ�����ݺͷ�Ӧ�Ļ�ѧ����ʽ���Լ�����ɴ�ʵ��������Ҫ����Ϊ80%�Ĵ�п��������
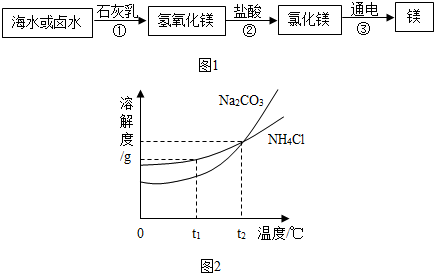
��� �⣺��1����ˮ�м������ˮʱ�������������ĭ�϶࣬����ˮ���������������������Ӳˮ��
�������ʱ��ˮ�еĸ����ӡ�þ�����ܹ���̼��Ƴ�����������þ��������ʽ��ˮ���������Ӷ�����ˮ��Ӳ�ȣ�
��2�����������У���Ԫ�صĻ��ϼ���-2�����ݻ�������Ԫ�ػ��ϼ۴�����Ϊ���֪����Ԫ�صĻ��ϼ���+4�����Ա�ʾΪ��$\stackrel{+4}{Cl}$O2��
��3��ij��ֳ�����ⶨˮ����ˮ�������ǿ������ѡ��pH��ֽ����ѡ��C��
��4����ѧ������ͼ1װ�����ⶨˮ����ɣ�����aΪ��Դ�ĸ�����
����ˮ��ˮ������Ϊx��
����������$\frac{0.3g}{0.3g+49.7g-x}$��100%=0.625%
x=2g
���Թ����ռ�������������������������������Ϊy��
2H2O$\frac{\underline{\;ͨ��\;}}{\;}$2H2��+O2����
36 32
2g y
$\frac{36}{2g}$=$\frac{32}{y}$
y��1.8g
��ʵ��õ�����ˮ����ɵĽ�����ˮ����Ԫ�غ���Ԫ����ɣ�
��5����ͨ������������ָ���������ÿ�֪��a������̨��b�Ǿƾ��ƣ�
�ڱ���ʵ������п�ͽ�Ũ�����ᷴӦ���õ������ȶ���������������Ӧ��ѡ�õķ���װ��Ϊ����п�����ᷴӦ�����Ȼ�п����������ѧ����ʽΪ��Zn+2HCl�TZnCl2+H2����
��ͼ��A��Bװ���зŵ�ҩƷ�ֱ�������������Һ��Ũ���ᣬ���������Ȼ������������������ѡ��c��
��ʵ�鿪ʼͨһ��ʱ���������Ŀ���dz���װ���еĿ�����ȷ�ϴ�Ŀ���Ѿ��ﵽ��ʵ���������Ϊ��Eװ��β���ռ������鴿����������Ѿ���������������ž���
�ݼ���һ��ʱ���C�з����ķ�Ӧ������ͭ�������ڼ��ȵ������·�Ӧ����ͭ��ˮ����ѧ����ʽΪ��CuO+H2$\frac{\underline{\;\;��\;\;}}{\;}$Cu+H2O��
ʵ�������ֹͣ���ȣ�����ͨ������Ŀ��֮һ�������ɵ�ͭ�����ȴ����һĿ��Ϊʹ��Ӧ���ɵ�ˮ����ȫ���գ�
��Eװ�õ�����Ϊ��ֹ�����е�ˮ�����Ͷ�����̼��Dװ�����գ�
����װ�ã��ᵼ��ˮ������ƫ�Ӷ�������Ԫ������ƫ��һ������ʵ���õ�����������ƫ��
�߷�Ӧ����ˮ������Ϊ��121.8g-120.0g=1.8g��
ˮ����Ԫ������Ϊ��1.8g��$\frac{2}{18}$��100%=0.2g��
ˮ����Ԫ������Ϊ��101.6g-100.0g=1.6g��
�����ʵ�鷽����õ�����������Ϊ��$\frac{0.2g}{1}$��$\frac{1.6g}{16}$=2��1��
��A������ͭû��ȫ������ԭ������Ӱ��ʵ�����������ԭ�Ӹ����Ȳⶨ��
B������ͭ�л�������������������Ӱ��ʵ�����������ԭ�Ӹ����Ȳⶨ��
C������ͭδ��ȫ����ᵼ��ʵ�����������ԭ�Ӹ�����ƫ��
D������ͭ�л�������������ͭʱ��������ͭ���ȷֽ���������ͭ��ˮ���ᵼ��ʵ�����������ԭ�Ӹ�����ƫ��
��ѡ��AB��
����μӷ�Ӧ��п����Ϊm��
��Zn+2HCl�TZnCl2+H2����CuO+H2$\frac{\underline{\;\;��\;\;}}{\;}$Cu+H2O
��֪��Zn��H2O��
Zn��H2O��
65 18
m 1.8g
$\frac{65}{m}$=$\frac{18}{1.8g}$
m=6.5g
������ʵ������Ҫ��п������Ϊ��$\frac{6.5g}{\frac{1}{5}}$=32.5g��
����ɴ�ʵ��������Ҫ����Ϊ80%�Ĵ�п����Ϊ��32.5g��80%=40.625g��
����ɴ�ʵ��������Ҫ����Ϊ80%�Ĵ�п����Ϊ40.625g��
�ʴ�Ϊ����1������ˮ����У�
��2��$\stackrel{+4}{Cl}$O2��
��3��c��
��4������1.8��ˮ������������Ԫ����ɵģ�
��5��������̨���ƾ��ƣ�
�ڱ���Zn+2HCl�TZnCl2+H2����
��C��
����Eװ��β���ռ����岢�鴿��
�ݣ������ɵ�ˮ����ȫ���գ�
��ֹ�����е�ˮ�Ͷ�����̼����װ��D��ƫ��
��2��1��
��AB��
��40.625�ˣ�
���� �������ʵ�飬��ѧ�ؽ���ʵ�顢����ʵ�飬�ǵó���ȷʵ����۵�ǰ�ᣬ���Ҫѧ�����ʵ�顢����ʵ�顢����ʵ�飬Ϊѧ�û�ѧ֪ʶ�춨������

 ���Ͱ�ͨ�������Сѧ��ʱͬ�����ϵ�д�
���Ͱ�ͨ�������Сѧ��ʱͬ�����ϵ�д� ���Ͱ�ͨ������ϵ�д�
���Ͱ�ͨ������ϵ�д� �ٷ�ѧ����ҵ��������ϵ�д�
�ٷ�ѧ����ҵ��������ϵ�д�| A�� | +4 | B�� | +5 | C�� | +6 | D�� | +7 |
| ���� | �� | �� | �� | �� |
| ��Ӧǰ������/g | 54 | 17 | 20 | 10 |
| ��Ӧ�������/g | 10 | 0 | 2 | x |
| A�� | xΪ89 | B�� | �μӷ�Ӧ�ı����ʵ�����Ϊ20 g | ||
| C�� | �÷�ӦΪ�ֽⷴӦ | D�� | ������ȫ����Ӧ�� |
���ϣ�����������茶��廯ѧʽΪFeSO4•��NH4��2SO4•6H2O����Է�������Ϊ392��������ˮ���������Ҵ���
��������ʵ��ܽ�ȣ���λ��g�������
| �¶�/�� | FeSO4 | ��NH4��2SO4 | FeSO4•��NH4��2SO4•6H2O |
| 10 | 20.0 | 73 | 17.2 |
| 20 | 26.5 | 75.4 | 21.6 |
| 30 | 32.9 | 78 | 28.1 |
ij��ȤС���ѧ���ú�������ͭ�ķ���м�Ʊ���������茶��壬������ͼ��
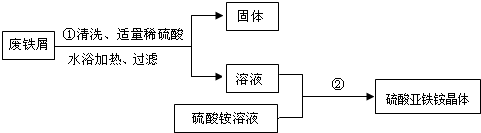
��1���������з�����Ӧ�Ļ�ѧ����ʽΪFe+H2SO4�TFeSO4+H2����
��2����������IJ����������ձ�����������©����
��3���������й���ʱ��Ҫ���Ƚ��е�ԭ���Ƿ�ֹ�¶Ƚ���FeSO4�������������������ʣ����ͻ�ʹFeSO4�����ܶ���ܽ⣬��߲��������ʣ���
��4�������ڵ�Ŀ���ǵõ���������茶��壮����˳����b��c��a��d��
a������ b������Ũ�� c����ȴ�ᾧ d��ϴ�Ӹ���
��5����������ʹ���Ҵ�ϴ�ӣ��ɿ������ɣ����������ŵ���AB��
A��������ˮϴ������ɵľ������
B���ƾ��ӷ����ɵ������ɾ���
��6����ʵ��ȡ����м15g�����յõ���������茶���39.2g����ԭ����м����Ԫ�ص���������������37.3����ȷ��0.1%����Ҫ��д���̣���
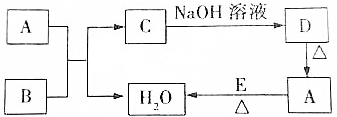
 ��ͼ�dz��л�ѧ�г������ʼ��ת����ϵ�����мס���Ϊ���ʣ�A��B��C��DΪ�������A��B�����Ԫ����ͬ��C��D�����Ԫ��Ҳ��ͬ�����෴Ӧ���������ַ�Ӧ������ȥ�����жϣ�
��ͼ�dz��л�ѧ�г������ʼ��ת����ϵ�����мס���Ϊ���ʣ�A��B��C��DΪ�������A��B�����Ԫ����ͬ��C��D�����Ԫ��Ҳ��ͬ�����෴Ӧ���������ַ�Ӧ������ȥ�����жϣ�