��Ŀ����
����2�����ڴ���ս��ʱ�ڣ��ҹ��Ϳ�ʼ������ʹ����������Ԫ1����ʱ�������ҹ��Ѿ��㷺ʹ�á���ͼΪ��ҵ��������Ҫ�豸——��¯������ش���������
��1�����ڵؿ��к��������н���Ԫ�صĵ� λ��
��2����¯���������У�CO������ʯ��Ӧ��Ҫ������¯������д��������(Fe2O3)��CO��Ӧ�Ļ�ѧ����ʽ___________________________��
��3������ʯ�ڸ�¯��ұ���õ����϶�̼�����ס���ȵ������������ĺ�̼����ΧΪ_________________��
�������ؿ���Ԫ�غ����ɶൽ�ٵ�˳��Ϊ�����衢����������ˣ����ڽ���Ԫ����ռ��2λ��������̼����2%~4.3%֮�䣬�ֵĺ�̼����0.03%~2%֮�䡣
�𰸣���1��2 ��2��3CO+Fe2O3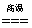 2Fe+3CO2 ��3��2%~4.3%
2Fe+3CO2 ��3��2%~4.3%

��ϰ��ϵ�д�
�����Ŀ