��Ŀ����
19��ijƷ�Ʋ��Ƽ�����Ҫ�ɷ���̼��ƣ����ʲ�����Ԫ�أ�����12g���ֲ��Ƽ���Ʒ������ϡ���ᣬ��������������������������±���ʾ�����ʿ�����ˮ���������ᷴӦ���������μ��������������Һ���Ȼ��Ƶ���������Ϊ20%�������������ݽ��м��㣮| ��һ�� | �ڶ��� | ������ | |
| ����ϡ���������/g | 18.25 | 36.5 | 47.9 |
| �������������/g | 2.2 | 4.4 | 4.4 |
��2������ϡ���������ʵ�����������
���� �ɱ������ݿ�֪��12g���ֲ��Ƽ���Ʒ��ϡ������ȫ��Ӧʱ���ɶ�����̼��������4.4g�����ݶ�����̼���������Լ���̼��Ƶ���������һ�����Լ�����ݲ��Ƽ����Ƶ�����������
��һ�μ���18.25gϡ����ʱ����̼��Ʒ�Ӧ���ɶ�����̼������Ϊ2.2g�����ݶ�����̼���������Լ���ϡ�������Ȼ������������һ�����Լ�������ϡ���������ʵ�����������
��� �⣺��1������Ʒ��̼��Ƶ�����Ϊx��
CaCO3+2HCl�TCaCl2+H2O+CO2����
100 44
x 4.4g
$\frac{100}{44}=\frac{x}{4.4g}$
x=10g��
̼����и�Ԫ������Ϊ��10g��$\frac{40}{100}$��100%=4g��
��ݲ��Ƽ����Ƶ���������Ϊ��$\frac{4g}{12g}$��100%=33.3%��
����ݲ��Ƽ����Ƶ���������Ϊ33.3%��
��2����18.25gϡ�������Ȼ��������Ϊy��
CaCO3+2HCl�TCaCl2+H2O+CO2����
73 44
y 2.2g
$\frac{73}{44}=\frac{y}{2.2g}$
y=3.65g
����ϡ���������ʵ���������Ϊ��$\frac{3.65g}{18.25g}��100%$=20%
������ϡ���������ʵ���������Ϊ20%��
���� ������Ҫ����ѧ�����ü��跨�ͻ�ѧ����ʽ���м�����ƶϵ�����������ʱҪע��淶�Ժ�ȷ�ԣ�

 ���Ŀ��ּ�����ҵ�����ҵ����������ϵ�д�
���Ŀ��ּ�����ҵ�����ҵ����������ϵ�д� ����ѵ��ϵ�д�
����ѵ��ϵ�д�| A�� | �ӵ�ʳ�� | B�� | Ǧ��о | C�� | �������� | D�� | �ƺ�ơ�� |
| A�� | ��BaCl2��Һ�м���Na2SO4 | B�� | ��ϡ�����м����ռ���Һ | ||
| C�� | �����ʯ��ˮ�м���ϡ���� | D�� | ��NaCl��Һ�м���CaCl2��Һ |
| A�� | ľ̿��������ȼ�շ����� | |
| B�� | ������ͭ��Һ�е����Ȼ�����Һ������ɫ��״���� | |
| C�� | ����������ȼ�շ�������ɫ���� | |
| D�� | ����ʯ���м�ˮ���������Ϸ����ִ������� |
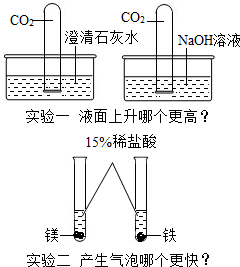 �����ߡ����졢��ǿ���İ��˾����ڻ�ѧѧϰ���������֣���������⣬��Ҫ��ش����⣺
�����ߡ����졢��ǿ���İ��˾����ڻ�ѧѧϰ���������֣���������⣬��Ҫ��ش����⣺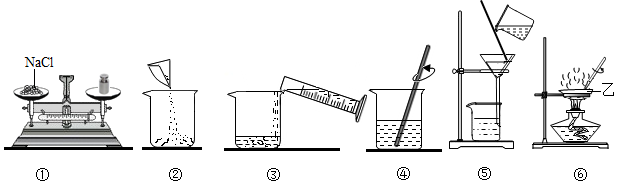
 ��ͼ��һ�ֿ����ڼ�����ϴ���ȵĶ��װ��
��ͼ��һ�ֿ����ڼ�����ϴ���ȵĶ��װ��