��Ŀ����
14��2015��1��9�գ�2014��ȹ��ҿ�ѧ������������ھ�¡�ؾ��У��״���CH3OH����ȡ��̼ϩ��������ù��Ҽ�������һ�Ƚ������й��ڼ״���˵������ȷ���ǣ�������| A�� | �״���̼Ԫ�ص���������Ϊ37% | |
| B�� | �״��ڿ�������ȫȼ�յIJ������Ҵ���ͬ����Ϊ������̼��ˮ | |
| C�� | һ���״�������һ��̼ԭ�ӡ��ĸ���ԭ�Ӻ�һ����ԭ�ӹ��� | |
| D�� | �״�����Է�������Ϊ32 |
���� A�����ݻ�������Ԫ�����������ļ��㷽����������
B�����������غ㶨����������
C�����ݼ״��Ļ�ѧʽ��������
D��������Է��������ļ��㷽����������
��� �⣺A���״��У�̼Ԫ�ص���������Ϊ��$\frac{12}{12+1��4+16}��100%$=37.5%���ʴ���
B���״�����̼���⡢������Ԫ����ɵģ���ȫȼ�յIJ�����ˮ�Ͷ�����̼������ȷ��
C���ɼ״��Ļ�ѧʽCH3OH��֪��һ���״���������һ��̼ԭ�ӡ��ĸ���ԭ�Ӻ�һ����ԭ�ӹ��ɵģ�����ȷ��
D���״�����ѧʽ��CH3OH������Է��������ǣ�12+1��4+16=32������ȷ��
��ѡA��
���� �����Ƕ����ʵĻ�ѧʽ���еĽ�ȫ��Ŀ��飬�漰����ѧʽ�Ķ��ּ��㣬���Ի�ѧʽ����������˷�����

��ϰ��ϵ�д�
�����Ŀ
4����������������ɻ�����Ⱦ���ǣ�������
| A�� | �����㵹���� | B�� | ������úȡů | ||
| C�� | ����̫���ܸ�·�Ʒ��� | D�� | �ù��ķϵ�����ⶪ�� |
2����ͼװ�ò�������֤�������غ㶨��ʵ����ǣ�������
| A�� | 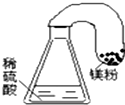 | B�� | 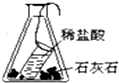 | C�� | 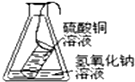 | D�� |  |
19������������ƿ�зֱ�ʢ�п������������������������ǵ���÷����ǣ�������
| A�� | �ô����ǵ�ľ������ƿ�� | B�� | ����ζ | ||
| C�� | ��������ʯ��ˮ | D�� | �ֱ��ȡ���Ե����� |
6��̽�������������������IJ��ʱ����ȼ�ŵ�ľ�����뼯��ƿ�У���Ҫ��Ϊ��֤����������
| A�� | ���������к��н϶������̼����ʹȼ�ŵ�ľ��Ϩ�� | |
| B�� | ���������к��н϶�ˮ��������ʹȼ�ŵ�ľ��Ϩ�� | |
| C�� | ������������������������������� | |
| D�� | ���������к��еĶ�����̼�����������еĶ�����̼�� |
3��ijͬѧд��O2��ѧ���ʵķ���ʽ�У���ȷ���ǣ�������
| A�� | O2+S�TSO2 | B�� | O2+2Cu$\frac{\underline{\;\;��\;\;}}{\;}$2CuO | ||
| C�� | 3O2+4Fe$\frac{\underline{\;��ȼ\;}}{\;}$2Fe2O3 | D�� | O2+2H2$\frac{\underline{\;ȼ��\;}}{\;}$2H2O |
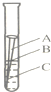 ���й����ϱ���������������ÿ����ʴ�����ϵĽ����豸�Ͳ����൱�����������20%��40%��ijͬѧΪ̽�������������������һ֧�ྻ������������װ������ˮ���Թ��У���ͼ����һ��ʱ���Ժ�������B����A��B��C�������ȳ������⣮��ʵ��˵������������ʵ������������е�ˮ������ �����˻�ѧ��Ӧ��
���й����ϱ���������������ÿ����ʴ�����ϵĽ����豸�Ͳ����൱�����������20%��40%��ijͬѧΪ̽�������������������һ֧�ྻ������������װ������ˮ���Թ��У���ͼ����һ��ʱ���Ժ�������B����A��B��C�������ȳ������⣮��ʵ��˵������������ʵ������������е�ˮ������ �����˻�ѧ��Ӧ�� С���Ӽ�������һƿ���õ�ҽ�ù���������Һ����ͬѧ��һ��ⶨ�����Ǵ�ƿ��ȡ������Һ��50g�����������������̣����������������뷴Ӧʱ��Ĺ�ϵ��ͼ��ʾ�������Һ������˫��ˮ�����������Ƕ��٣�
С���Ӽ�������һƿ���õ�ҽ�ù���������Һ����ͬѧ��һ��ⶨ�����Ǵ�ƿ��ȡ������Һ��50g�����������������̣����������������뷴Ӧʱ��Ĺ�ϵ��ͼ��ʾ�������Һ������˫��ˮ�����������Ƕ��٣�