��Ŀ����
�ճ�ʹ�õĽ������ϣ���������ںϽ𡣻�ͭ����п����Ҫ����Ԫ�ص�ͭ�Ͻ�
(1)��ͭ(Cu2O)���ҹ��Ŵ���ȡ��ͭ��һ��ԭ�ϡ�Cu2O��ͭԪ������Ԫ�ص���������
.��
(2)���ǵĻ�ֻͭ����п��ͭ����22 g���Ƿ���ʢ��100 gϡ������ձ���(��������)�������Dz����ܽ���ձ��л�����������121. 8 g������:
�ٲ���������������
�ڵ�����ͭ��������
�۷�Ӧ��������Һ��ZnSO4����������(����������һλС��)��
��1��8:1
��2���ٲ�������������Ϊ22g+100g-121.8g=0.2g
���赯����п������Ϊx,���ɵ�ZnSO4����Ϊy
Zn + H2SO4=ZnSO4 + H2��
65 161 2
X y 0.2g
65:2=x: 0.2g x=6.5g
161:2=y:0.2g y=16.1g
������ͭ������Ϊ22g-6.5g=15.5g
��Ӧ��������Һ����Ϊ121.8g-15.5g=106.3g
��6.5g+100g-0.2g=106.3g
�۷�Ӧ��������Һ��ZnSO4����������Ϊ 16.g/106.3g��100%=15.

��ʡ��������ú̿��Դ�ḻ����˹������ú�㼰��Χ�Ҳ��У��Ǿ����к�������ܳƣ���Ҫ�ɷ��Ǽ��顣
(1)����д��������ȫȼ�յĻ�ѧ����ʽ��__________________________________��
(2)ú����˹��ը��������Ҫ������
����˹�����ڱ�ը���ķ�Χ�ڣ���________________________________________��
| ��ȼ���� | ��ը���ޣ���������� |
| H2 | 4.0%~74.2% |
| CH4 | 5%~15% |
| CO | 12.5%~74.2% |
(3)�ұ��dz�����������ı�ը���ޣ�����ݴ��жϣ�
����������ը��������____��
(4)����ͼ���У���ȼ�պͱ�ը�ص���

A B C D
(5)ij�ִ����Ŀ����ˡ���Ũ����˹���缼��������Ч����˿������ܱߵ����������õ硣���ּȲ�ú�ַ��������������ŵ���___________________________________��
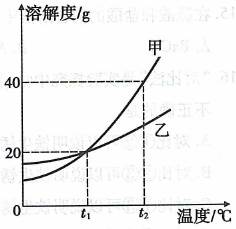 ��ʾ��
��ʾ��