��Ŀ����
11��ʵ����ʹ�ÿ�״ʯ��ʯ��CaCO3����ϡ������CO2�����۲첻�������ݲ�������������ʣ��ʱ��ͨ����Ϊ��Ӧ������ʣ��Һ�����ԣ�ij��ȤС��Դ˽���������̽������ʵ�������
��1�����Թ��м���5��ʯ��ʯ���㵹Լ�Թ����1/3��ϡ���ᣨ1��1�����۲첻�����ݺ����й���ʣ�ࣻ���Թܾ��ã�
��2��ȡ�ϲ���Һ����pH��ֽ�����ҺpH=2��˵����Һ�����ԣ�pH�ⶨ�У����ϲ���Һ�ε���ֽ�ϡ��ĺ�һ�������ǰ���ֽ�ͱ���ɫ�����գ�
��ʵ��˼����Ӧʣ��Һ������Щ���ʣ�������ʹ��ҺpH=2������Щ��
��ʵ��̽����
��3����ͬѧͨ��ʵ���ų���CaCl2�Ŀ����ԣ�������ʵ�鷽����ȡ�����Ȼ�����Һ���Թ��У�������Һ��pH��
��4����ͬѧ��CO2ͨ������ˮ�������ͣ����pH��5��
��5���ۺϼס�����ͬѧʵ�飬�ó��Ľ����ǣ�ʣ��Һ�л��������ᣮ
��ʵ��˼�����Թ���ʣ������Ƿ��Ժ���CaCO3��
��ʵ��̽����
С��ͬѧ���Թ��м�������ϡ���ᣨ1��1�������д������ݲ�����
��6���ɴ˵ó��Ľ������Թ���ʣ������Ժ���̼��ƣ�д����Ӧ�Ļ�ѧ����ʽ��CaCO3+2HCl�TCaCl2+H2O+CO2����
��7���Ƚ�ϡ�������ǰ���ʵ�������͡����д������ݲ�������ԭ�������Ũ������
����չ���졿
��8����Ӧʣ��Һ����һ��ʱ�����ҺpH��������Ҫԭ����̼��ƺ����������Ӧ
��9��������100g 14.6%��ϡ�����������ʯ��ʯ��Ӧ�����������ݲ���ʱ��ֹͣ�ռ����壮�ռ�����CO2��������ѡ���������������=����8.8g��
���� �����Ե���Һ��pHС��7���ⶨ��ҺpH�ķ����ǣ��ýྻ����IJ�����պȡ������ķ�ˮ������pH��ֽ�ϣ��ѱ�ɫ��pH��ֽ�����ɫ�����գ����ɵ���Һ��pH������Ũ��Խ��̼��Ʒ�Ӧ��Խ�죮
��� �⣺��ʵ�������
��2��ȡ�ϲ���Һ����pH��ֽ�����ҺpH=2��˵����Һ�����ԣ�
pH�ⶨ�У����ϲ���Һ�ε���ֽ�ϡ��ĺ�һ�������ǰ���ֽ�ͱ���ɫ�����գ�
��������ֽ�ͱ���ɫ�����գ�
��ʵ��̽����
��3��ͨ��ʵ���ų���CaCl2�Ŀ����ԣ���ʵ�鷽���ǣ�ȡ�����Ȼ�����Һ���Թ��У���pH��ֽ�ⶨ��Һ��pH��
���ȡ�����Ȼ�����Һ���Թ��У�������Һ��pH��
��5���ۺϼס�����ͬѧʵ�飬�ó��Ľ����ǣ�ʣ����Һ�л��������ᣮ
������ᣮ
��ʵ��̽����
��6��С��ͬѧ���Թ��м�������ϡ���ᣨ1��1�������д������ݲ������ɴ˵ó��Ľ������Թ���ʣ������Ժ���̼��ƣ�̼����������ᷴӦ�����Ȼ��ơ�ˮ�Ͷ�����̼��
����Թ���ʣ������Ժ���̼��ƣ�CaCO3+2HCl�TCaCl2+H2O+CO2����
��7���Ƚ�ϡ�������ǰ���ʵ���������д������ݲ�������ԭ��������Ũ����������̼��Ʒ�Ӧ��������Ũ������
����չ���졿
��8����Ӧʣ��Һ����һ��ʱ�����ҺpH��������Ҫԭ����̼��ƺ����������Ӧ��������٣����Լ�����pH���ߣ�
���̼��ƺ����������Ӧ��
��9��������100g14.6%��ϡ�����������ʯ��ʯ��Ӧʱ�����ݷ�Ӧ�Ļ�ѧ����ʽ�����֪�����ɶ�����̼��������8.8g�����ڿ��������ݲ���ʱ����û����ȫ��Ӧ�������ռ�����CO2������8.8g�������
�������
���� ʵ������������֮������õ����ڱ��֣����Ҫѧ�����ʵ�顢�۲�ʵ�顢����ʵ�飬Ϊ��ʾ����֮������õ�ʵ�ʵ춨������

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�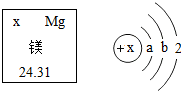 ��ͼ��þԪ�غ�þԭ�ӵ������Ϣ������˵���д�����ǣ�������
��ͼ��þԪ�غ�þԭ�ӵ������Ϣ������˵���д�����ǣ�������| A�� | þԪ�ص����ԭ��������24.31 g | |
| B�� | ��þԭ�ӽṹʾ��ͼ��a=2��b=8 | |
| C�� | þԭ���У�������=�˵����=������=12 | |
| D�� | þԭ���ڻ�ѧ��Ӧ����ʧȥ2�������γ�Mg2+ |
| ������� | �������� | ��ȥ���ʵ��Լ��� | |
| A | NaCl���� | Na2CO3 | ����ϡ���ᣬ���� |
| B | CO | CO2 | ��������Ũ��Һ������ |
| C | KCl | KClO3 | MnO2������ |
| D | FeSO4��Һ | CuSO4 | �������ۣ����� |
| A�� | A | B�� | B | C�� | C | D�� | D |
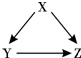 X��Y��ZΪ���л�ѧ���������ʣ�����֮�������ͼ��ʾ��ת����ϵ����һ�������¾���һ��ʵ�֣���
X��Y��ZΪ���л�ѧ���������ʣ�����֮�������ͼ��ʾ��ת����ϵ����һ�������¾���һ��ʵ�֣���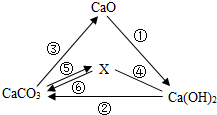 �й�����֮��IJ���ת����ϵ��ͼ��ʾ�����С�-����ʾ����֮���ܷ�����ѧ��Ӧ����������ʾ����֮���ת����ϵ����ش�������⣺
�й�����֮��IJ���ת����ϵ��ͼ��ʾ�����С�-����ʾ����֮���ܷ�����ѧ��Ӧ����������ʾ����֮���ת����ϵ����ش�������⣺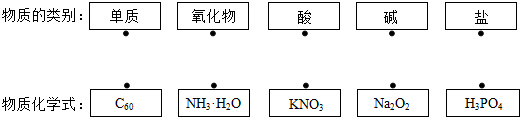
 ���������뿪������
���������뿪������