��Ŀ����
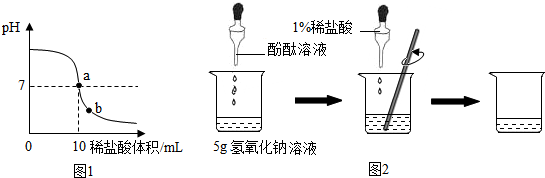
20��ijͬѧ���ⶨNa2CO3��NaCl����������Na2CO3�������������ֱ�ȡag��Ʒ�������з�������ʵ�飬�������ǣ�������| A�� | ������ϡ�����ַ�Ӧ������ˮ���ռ���VmL���� | |
| B�� | ������ϡ�����ַ�Ӧ��������Ӧ�����Һ������Ϊm1g | |
| C�� | ������Ũ�����ַ�Ӧ���ݳ���������NaOH�������գ�������������m2g | |
| D�� | ������Ca��OH��2��Һ��ַ�Ӧ�����������ˡ�ϴ�ӡ�����õ�m3g���� |
���� �������е�֪ʶ���з�����Ҫ�ⶨ�������̼���Ƶĺ�������������̼���Ƶ����ʣ����ᷴӦ���ⶨ���ɵ�������������Dzⶨʣ�����ʵ�������Ҳ�������������Ʒ�Ӧ���ⶨ���ɳ�����������Ȼ����㣬�ݴ˽�ɣ�
��� �⣺
A�����ڶ�����̼��������ˮ�����ʣ�ͨ����ˮ���ռ��õ��Ķ�����̼�����С��ʵ�����ɵĶ�����̼���������A��������
B��Ҫ����ʣ����Һ���������м��㣬������֪���μӷ�Ӧ��ϡ��������������������㣬��B��������
C��ʹ��Ũ���ᷴӦ������Ũ������лӷ��ԣ���ӷ����Ȼ������壬����ʹ���������ƽ�������ʱ��������̼Ҳ�����������Ʒ�Ӧ���ʸ��ݹ�����������������̼���Ƶĺ�������C��������
D��������Ca��OH��2��Һ��ַ�Ӧ�����������ˡ�ϴ�ӡ�����õ�m2g���壬�˹���������������ɵ�̼��Ƶ�����������̼��ƿ�������̼���Ƶ���������������̼���Ƶ�������������D������
��ѡD��
���� ���⿼���˳��������ɷֵIJⶨ����ɴ��⣬�����������ʵ����ʽ��У�

��ϰ��ϵ�д�
 ��Կ���Ծ�ϵ�д�
��Կ���Ծ�ϵ�д�
�����Ŀ
15������˵���д�����ǣ�������
| A�� | ����Ǽ� | B�� | �ɱ����DZ� | C�� | ʳ�β����� | D�� | ���ʯ������ |
10������ͼ������ȷ��ӳ���Ӧ��ϵ���ǣ�������
| A�� | 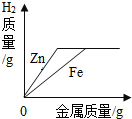 �����������Ũ�ȵ�ϡ�����зֱ���������Zn��Fe | |
| B�� | 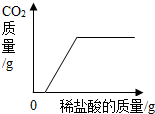 ��������NaOH��Na2CO3��Һ����μ���ϡ���� | |
| C�� | 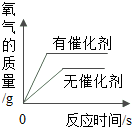 �õ���������Ũ�ȵĹ���������Һ���С�������������ȡ���� | |
| D�� |  һ���¶�ʱ����һ�����ı���ʯ��ˮ�м�����ʯ�� |
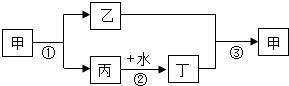 �ס��ҡ���������Ϊ��ѧ��ѧ�еij������ʣ����м��Ǵ���ʯ����Ҫ�ɷ֣������������ЧӦ����Ҫ���壬����֮���ת����ϵ��ͼ������������Ϣ�ش����⣺
�ס��ҡ���������Ϊ��ѧ��ѧ�еij������ʣ����м��Ǵ���ʯ����Ҫ�ɷ֣������������ЧӦ����Ҫ���壬����֮���ת����ϵ��ͼ������������Ϣ�ش����⣺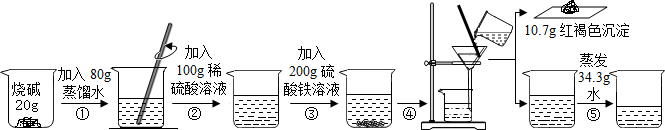

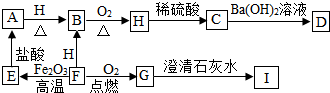
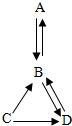 A��B��C��D������ͬһ��Ԫ�أ����ǵ�ת����ϵ��ͼ���������ʺͷ�Ӧ��������ȥ����
A��B��C��D������ͬһ��Ԫ�أ����ǵ�ת����ϵ��ͼ���������ʺͷ�Ӧ��������ȥ����