��Ŀ����
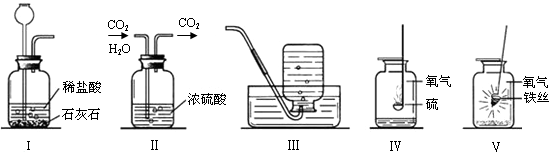
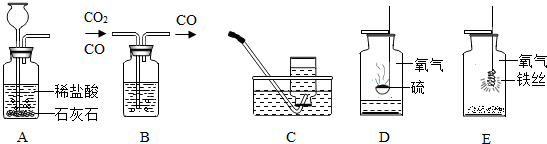
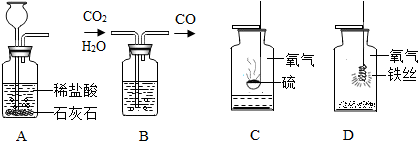
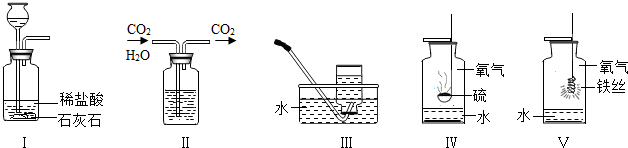
�ü���ƿ����ɶ���ʵ�顣���������ʵ��ʾ��ͼ���ش��й����⣺
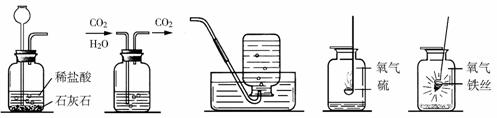
I II III IV ��
��1��ʵ��I��ɵ�ʵ����__________________��ʵ���ļ���ƿ��װ����Լ���______________��ʵ��III�ռ���������������ʵ������ȡ�����Ļ�ѧ����ʽ�� ___________________��
��2��ʵ����ļ���ƿ��Ԥ�ȼ���������ˮ����������______________________________��
��3��ʵ����۲쵽��������______________________________________________����Ӧ�Ļ�ѧ����ʽ��____________________________________________��
��4������ɢ���������ʵ��ʱ��Ϊ�˱�������ƿ������Ӧ��ע��IJ����� ��
��1����ȡ������̼���� Ũ���ᣨ��ŨH2SO4�� 2KClO3====2KCl+3O2��
(��2KMnO4 �� K2MnO4+MnO2+O2�� �� 2H2O2 MnO2 2H2O+ O2�� ) ��2�����ն�������ֹ��Ⱦ������������SO2����3���ٻ�����������ɺ�ɫ����۷ų����� 3Fe+2O2 ��ȼ Fe3O4 (4) ȼ�ŵ����ʲ��Ӵ�����ƿ�ڱڣ�����ʵ����ļ���ƿ�ڼ�����ϸɳ��ˮ��

 ����ͼ���������������ϵ�д�
����ͼ���������������ϵ�д� ����ѧҵ���Ե�����ϵ�д�
����ѧҵ���Ե�����ϵ�д�