��Ŀ����
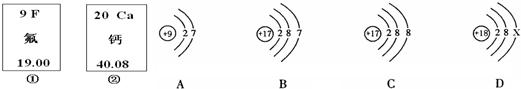
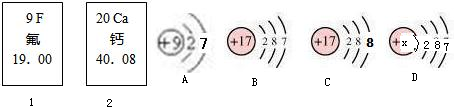
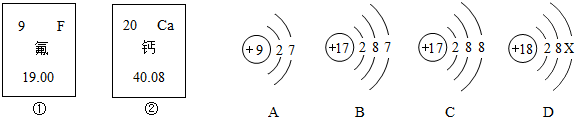
��ͼ�еĢ٢��Ƿ�Ԫ�ء���Ԫ����Ԫ�����ڱ��е���Ϣ��A��B��C��D���������ӵĽṹʾ��ͼ��

����ش�
(1)��Ԫ�ص����ԭ������Ϊ__________����Ԫ�ص�ԭ������Ϊ__________��
(2)x=____��
(3)A��B��C��D������ͬ��Ԫ�ص�������_________������ţ���
(4)A���ӵĻ�ѧ������B��C��D����һ�����ӵĻ�ѧ����_________������ţ���
(5) 2008��5�£�ij�ط����������(HF)й©��������Ա����ʯ�Ҷ�й©�ص���д�������д����ѧ����ʽ_____________________________��
(1)��Ԫ�ص����ԭ������Ϊ__________����Ԫ�ص�ԭ������Ϊ__________��
(2)x=____��
(3)A��B��C��D������ͬ��Ԫ�ص�������_________������ţ���
(4)A���ӵĻ�ѧ������B��C��D����һ�����ӵĻ�ѧ����_________������ţ���
(5) 2008��5�£�ij�ط����������(HF)й©��������Ա����ʯ�Ҷ�й©�ص���д�������д����ѧ����ʽ_____________________________��
(1)19.00��20
(2)8
(3)BC
(4)B
(5)Ca(OH)2+2HF==CaF2+ 2H2O
(2)8
(3)BC
(4)B
(5)Ca(OH)2+2HF==CaF2+ 2H2O

��ϰ��ϵ�д�
 ���ſ����ϵ�д�
���ſ����ϵ�д� ���Ŀ����ϵ�д�
���Ŀ����ϵ�д� ������ӱ������ͯ������ϵ�д�
������ӱ������ͯ������ϵ�д�
�����Ŀ