��Ŀ����
8�������ǵ���Ҫ�ɷ���̼��ƣ�Ϊ�˲ⶨ��������̼��Ƶĺ�����С����ȡ30g������鼦���Ƿ����ձ��У��������м�����80gϡ����ǡ����ȫ��Ӧ�����輦�����г�̼�����������ɷֶ�������ˮ���Ҳ���ϡ���ᷴӦ������Ӧ���ձ������ʵ�������Ϊ101.2g������������м��㣺��1����ȫ��Ӧ�����ɶ�����̼������Ϊ8.8g
��2��̼��Ƶ�������
��3����̼���ǡ����ȫ��Ӧʱ������Һ�����ʵ��������������������һλС����
���� ��1��̼��������ᷴӦ�ų�������̼��ʹ��Ӧ��������С�����������غ㶨�ɿɼ���ų�������̼��������
��2�����ݷ�Ӧ�Ļ�ѧ����ʽ���ɷų�������̼���������㵰��������̼��Ƶ�������
��3�����ݷ�Ӧ�Ļ�ѧ����ʽ���ɷų�������̼���������㵰���������Ȼ��Ƶ�������Ȼ���������ʵ���������ȥ���ʵ����������Һ���������Ӷ����������Һ�����ʵ�����������
��� �⣺��1�����������غ㶨�ɣ����ɶ�����̼������Ϊ80g+30g-101.2g=8.8g�����8.8��
��2����30g��������̼��Ƶ�����Ϊx�������Ȼ��Ƶ�����Ϊy��
CaCO3+2HCl=CaCl2+H2O+CO2��
100 111 44
x y 8.8g
$\frac{100}{x}=\frac{111}{y}=\frac{44}{8.8g}$
x=20g
y=22.2g
��3��������Һ�����ʵ���������=$\frac{22.2g}{101.2g-��30g-20g��}$��100%��24.3%
�𣺣�2��30g��������̼��Ƶ�����Ϊ20g����3��������Һ�����ʵ���������Ϊ24.3%��
���� ���������غ㶨�������Ӧ�ų�������̼���������ǽ��к������Ļ��������ֳ�����֪ʶ���������������

| A�� | ���� | B�� | ������̼ | C�� | ���� | D�� | ϡ������ |
| A�� | N2O | B�� | NO2 | C�� | NH3 | D�� | N2O5 |
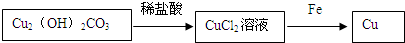 ��
��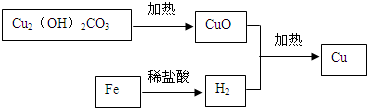 ��
��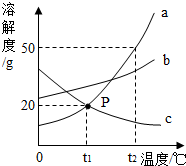 ��ͼ��a��b��c�������ʵ��ܽ�����ߣ�a��c���ܽ�������ཻ��P�㣮��ͼ�ش�
��ͼ��a��b��c�������ʵ��ܽ�����ߣ�a��c���ܽ�������ཻ��P�㣮��ͼ�ش�