��Ŀ����
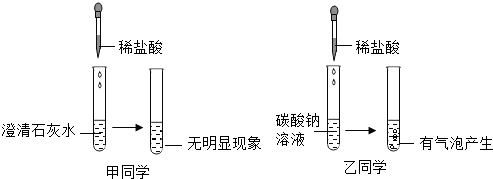
5��2015��3��22���ǵڶ�ʮ���조����ˮ�ա����ҹ��������ˮ�ա������������Ϊ����Լˮ��Դ������ˮ��ȫ��������ˮ�����ǵ��ճ����������������1�������п��÷���ˮ���������ˮ��Ӳˮ������ˮ����������еķ�����������ˮ��Ӳ�ȣ���ɱ��������
��2���ⶨ����ˮ�����ȳ���PH��ֽ�����������Ϊ���ڰ״ɰ��Ϸ�һСƬPH��ֽ���ýྻ�IJ�������ȡ����Һ����ֽ�ϣ����ձ�ɫ����������ֵ��
���� ��1������ˮ����������ˮ��Ӳˮ��
��2��������Һ�����Ⱦ���ⶨ�������ø���IJ�����պȡ����ͷ�ι���ȡ�������Ĵ�����Һ�������ڷ��ڸ���IJ������״ɰ��ϵĸ���pH��ֽ�ϣ��ٰ���ֽ��ʾ����ɫ�����ɫ���Ƚϣ����ɵó�������Һ��pH���
��� �⣺��1��Ӳˮ�������ˮ�����ɵ���ĭ���٣���ˮ�������ˮ�����ɵ���ĭ�϶࣬���Կ����÷���ˮ������ˮ��Ӳˮ�����ü�����еķ������ȿɽ���ˮ��Ӳ�ȣ�����ɱ��������
��2���ⶨ����ˮ�����ȳ���PH��ֽ�����������Ϊ���ڰ״ɰ��Ϸ�һСƬPH��ֽ���ýྻ�IJ�������ȡ����Һ����ֽ�ϣ����ձ�ɫ����������ֵ��
�ʴ�Ϊ����1������ˮ����У���2�����ձ�ɫ����������ֵ��
���� ˮ��Դ�����úͱ������ִ�������ٵ���Ҫ���⣬ͬʱҲ�ͳ�Ϊ�п�����Ҫ����֮һ��

��ϰ��ϵ�д�
�����Ŀ
13�������й���Һ��˵������ȷ���ǣ�������
| A�� | ��Һ�����ʺ��ܼ���ɣ�����ÿ����Һ��ֻ���������� | |
| B�� | ��������Һ�ܵ�������Ϊ��Һ�к��н϶������ƶ������� | |
| C�� | ����0.09%��������ˮ��һ�㲽���Ǽ��㡢�������ܽ� | |
| D�� | ��ϡ��Ũ����ʱ��һ��Ҫ��Ũ��������������ע��ˮ�� |
20����ͼ��ij�ܱ����������ʱ仯����ʾ��ͼ��������̼ԭ�ӣ����𡱴�����ԭ�ӣ��������й�˵��������ǣ�������
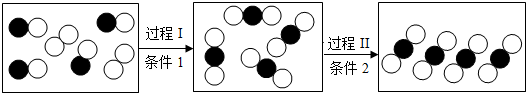

| A�� | �μӷ�Ӧ�����ʷ��Ӹ�����Ϊ1��1 | B�� | �����ٽ��ܵķ���֮��Ҳ�м�϶ | ||
| C�� | ���̢�Ӧ����Ϊ���Ϸ�Ӧ | D�� | ���̢����������仯 |
17�����ʯ��ʯī�����������кܴ���죬��ԭ���ǣ�������
| A�� | �������ǵ�ԭ�Ӵ�С��ͬ | |
| B�� | �������ǵ�ԭ����Ŀ��ͬ | |
| C�� | ���ʯ��ʯī�ɲ�ͬ��ԭ�ӹ��� | |
| D�� | ���ʯ��ʯī��̼ԭ�ӵ����з�ʽ��ͬ |
10������ɫ��ѧ����ָ��������
| A�� | ��ɫΪ��ɫ����������Ʒ | |
| B�� | ��������ԭ�ϡ����������������IJ�Ʒ | |
| C�� | ���Բ����κλ�ѧԪ�ص�����Ʒ | |
| D�� | ֻ��ʹ�ã����������IJ�Ʒ |