��Ŀ����
11����1����ҽ���á��������鷨�������鶯���Ƿ���˪��R���ж����仯ѧԭ��Ϊ��R+8HCl+4Zn�T4ZnCl2+3H2O+2As+H2��������˪�Ļ�ѧʽΪAs2O3����˪���������ѡ����ʡ��������������������2����һЩ���ڳ�ʹ��һ�ֱ�Яʽ���������ʽ¯����ʹ�õ�ȼ����Һ������������֪����Ļ�ѧʽ��C4H10����д������ȼ�յĻ�ѧ����ʽ��2C4H10+13O2$\frac{\underline{\;��ȼ\;}}{\;}$8CO2+10H2O��
���� ��1����������������鷨ԭ��ΪR+8HCl+4Zn�T4ZnCl2+3H2O+2As+H2�������û�ѧ��Ӧǰ��ԭ�ӵ���������Ŀ�������ı����ƶ���˪�Ļ�ѧʽ��
��2����д��ѧ����ʽʱ��������Ϥ��Ӧ�������ͷ�Ӧ�������������ݿ���ʵ�����������غ㶨�ɣ�
��� �⣺��1���ɻ�ѧ����ʽR+8HCl+4Zn�T4ZnCl2+3H2O+2As+H2����
���Կ���Zn��Cl��H����Ԫ�ص�ԭ���ڷ�Ӧǰ��ĸ�����ȣ�
�ڷ�Ӧ����2��Asԭ�ӣ�3��Oԭ�ӣ�����������غ㶨�ɿ�֪
R����As��O����Ԫ�أ���1����������2��Asԭ�Ӻ�3��Oԭ�ӣ�
����˪�Ļ�ѧʽΪAs2O3�����������
��2�������������ڵ�ȼ��������ȫ��Ӧ���ɶ�����̼��ˮ�������÷�Ӧ�Ļ�ѧ����ʽΪ��2C4H10+13O2$\frac{\underline{\;��ȼ\;}}{\;}$8CO2+10H2O��
�𰸣���1��As2O3�� �����
��2��2C4H10+13O2$\frac{\underline{\;��ȼ\;}}{\;}$8CO2+10H2O��
���� ��������Ϣ�ķ�ʽ�������ʻ�ѧʽ���ƶϣ�ѧ��Ӧ�������غ�ķ�����������ѧ��Ӧ�����ܶ����ʵĻ�ѧʽ������ȷ���ƶϣ���Ҫע�ⷽ���Ĺ����ܽᣮ

��ϰ��ϵ�д�
�����Ŀ
17��������10%NaCl��Һ�Ĺ����У�������Һ��NaCl��������С��10%�Ŀ���ԭ���ǣ�������Ͳ��ȡˮʱ���Ӷ��� ��������Һʱ�ձ���������ˮ��ʪ ������ƽ�����̳����Ȼ���ʱ�����벻����λ�þ͵�����ƽƽ�⣬Ȼ�������ƶ������� ��ʢװ��Һ���Լ�ƿ������ˮ��ϴ��������
| A�� | �٢ڢۢ� | B�� | �٢ڢ� | C�� | �٢ڢ� | D�� | �ڢۢ� |
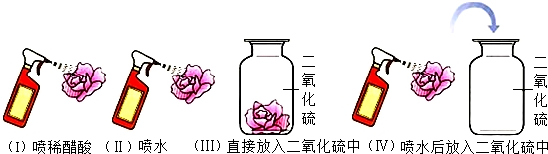
18������ʵ�������Ӧ�Ľ�����ȷ�ģ�������
| ѡ�� | A | B | C | D |
| ʵ�� |  | 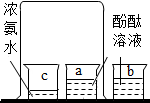 | 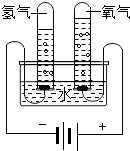 | 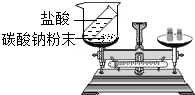 |
| ʵ������ | �ձ��ڱڸ��ŵij���ʯ��ˮ����� | a�ձ�����Һ����ɫ��ɺ�ɫ | �Թ�������������������������������������Ϊ2��1 | ��Ӧ����ƽָ������ƫ�� |
| ʵ����� | ˵������ȼ�ղ���������̼��ˮ | ��˵�������ڲ��ϵ��˶�����˵��Ũ��ˮ��ʹ��̪��� | ��˵��ˮ����Ԫ�ء���Ԫ����ɣ���˵��ˮ�����к�������Ӻ������� | ������̼���Ƶķ�Ӧ�����������غ㶨�� |
| A�� | A | B�� | B | C�� | C | D�� | D |
6��Ҫ���������ЧӦ�����⣬�����������ǿ��Բ�ȡ�Ĵ�ʩ��ȷ���ǣ�������
�ٽ�ֹʹ�û�ʯȼ��
�ڴ���ֲ���ֲݣ��Ͻ��ҿ��ķ�
�����ռ�̫���ܵ������Դ
�ܴ����ƹ��Ҵ����ͣ�
�ٽ�ֹʹ�û�ʯȼ��
�ڴ���ֲ���ֲݣ��Ͻ��ҿ��ķ�
�����ռ�̫���ܵ������Դ
�ܴ����ƹ��Ҵ����ͣ�
| A�� | �٢� | B�� | �ڢ� | C�� | �ڢۢ� | D�� | �٢ڢۢ� |
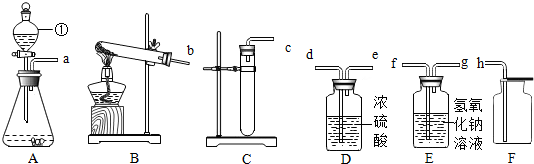
20��ʵ�����ø��������������������ˮ���ռ��������²�������װ��ҩƷ �ڼ�������� ���ռ����� �ܼ��� ��Ϩ��ƾ��� ��ȥ�����ܣ���ȷ�IJ����ǣ�������
| A�� | �٢ڢۢܢݢ� | B�� | �ڢ٢ܢۢݢ� | C�� | -�ڢ٢ܢۢޢ� | D�� | �٢ڢܢۢޢ� |