��Ŀ����
12��ij�������ŷŵķ�ˮ�к���̼��غ��������أ���ѧ��ȤС��Ϊ̽����ˮ��̼��صĺ�������ȡ50g��ˮ��Ʒ���ձ��У������ϡ����ǡ����ȫ��Ӧ����¼�й����������| ��Ӧǰ | ��Ӧ�� | |
| �ձ��ͷ�ˮ������ | ������������Ϊ9.8%������ | �ձ�����Һ������ |
| 60g | 75g | 132.8g |
��2���÷�ˮ��̼��ص�����������д��������̣���
��3����Ӧ����������ص�����Ϊ13.05g��
���� ��1����Ӧǰ�����ʵ�������֮��Ϊ������̼��������
��2�����ݶ�����̼��������ϻ�ѧ����ʽ�������ˮ��̼��ص�����������������������
��3�������������������������
��� �⣺��1�����ɶ�����̼������Ϊ��60g+75g-132.8g=2.2g��
���2.2g��
��2�����ˮ��̼��ص���������Ϊx��
K2CO3+H2SO4�TK2SO4+H2O+CO2��
138 44
50g��x 2.2g
$\frac{138}{44}=\frac{50g��x}{2.2g}$
x=13.8%
�𣺷�ˮ��̼��ص���������Ϊ13.8%��
��3�������е������ȫ��ת�Ƶ�������У���������ص�����Ϊy
H2SO4 ����K2SO4
98 174
75g��9.8% y
$\frac{98}{174}=\frac{75g��9.8%}{y}$
y=13.05g
���13.05��
���� �����Ĺؼ������������غ㶨�ɺ����û�ѧ����ʽ���㣮

��ϰ��ϵ�д�
 ȫ�ų��100��ϵ�д�
ȫ�ų��100��ϵ�д� Ӣ�ŵ��ϵ�д�
Ӣ�ŵ��ϵ�д�
�����Ŀ
2��������Сˬ�ܽ�İ�ȫ��ʶ�����д�����ǣ�������
| A�� | ú��й©__ �Ͻ����ص��� | B�� | ϡ��Ũ����--��ˮ����Ũ������ | ||
| C�� | �ڼ���վ����--��ֹ���� | D�� | ��������ֳ� ��ʪë����ס�ڱ� |
3����������������Ч���ƴ����������ǣ�������
| A�� | ����������ʹ���ܹ���PM2.5�Ŀ��� | |
| B�� | ��ʯ�Ҷ�úȼ�պ��γɵ��������������� | |
| C�� | ��������̫���ܡ����ܡ����ܵ������Դ | |
| D�� | �о����ۡ���Ч�Ĵ������Խ�������β�����к����ʵĺ��� |
20������ʵ����Ʒ����������ǣ�������
| A�� | ��̼��غ�������þ�Ʊ��������� | |
| B�� | ���Ȼ�����Һ�������������Ƿֱ��� | |
| C�� | ������������Һ��������李�����þ�������� | |
| D�� | ��ˮ��������粒�����������ƹ��� |
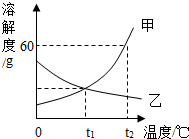 ��ͼ�Ǽס������ֹ������ʵ��ܽ�����ߣ�
��ͼ�Ǽס������ֹ������ʵ��ܽ�����ߣ�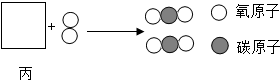
 ��
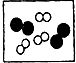
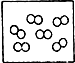
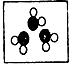
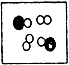
�� �ֱ��ʾ���ֲ�ͬԪ�ص�ԭ�ӣ����б�ʾ��������ǣ�������
�ֱ��ʾ���ֲ�ͬԪ�ص�ԭ�ӣ����б�ʾ��������ǣ�������